Advertisements
Advertisements
Question
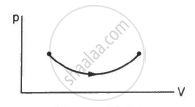
Consider the process on a system shown in figure. During the process, the work done by the system ______________ .
Options
continuously increases
continuously decreases
first increases then decreases
first decreases then increases
Advertisements
Solution
continuously increases
Work done by a system, `W=intPdV`
Here,
P = Pressure on the system
dV = change in volume.
Since dV is positive, i.e. the volume is continuously increasing, work done by the system also continuously increases.
APPEARS IN
RELATED QUESTIONS
Should the internal energy of a system necessarily increase if its temperature is increased?
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
The final volume of a system is equal to the initial volume in a certain process. Is the work done by the system necessarily zero? Is it necessarily nonzero?
Can work be done by a system without changing its volume?
The pressure p and volume V of an ideal gas both increase in a process.
(a) Such a process is not possible.
(b) The work done by the system is positive.
(c) The temperature of the system must increase.
(d) Heat supplied to the gas is equal to the change in internal energy.
In a process on a system, the initial pressure and volume are equal to the final pressure and volume.
(a) The initial temperature must be equal to the final temperature.
(b) The initial internal energy must be equal to the final internal energy.
(c) The net heat given to the system in the process must be zero.
(d) The net work done by the system in the process must be zero.
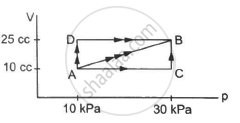
Figure shows three paths through which a gas can be taken from the state A to the state B. Calculate the work done by the gas in each of the three paths.
A substance is taken through the process abc as shown in figure. If the internal energy of the substance increases by 5000 J and a heat of 2625 cal is given to the system, calculate the value of J.

A mixture of fuel and oxygen is burned in a constant-volume chamber surrounded by a water bath. It was noticed that the temperature of water is increased during the process. Treating the mixture of fuel and oxygen as the system,
- Has heat been transferred?
- Has work been done?
- What is the sign of ∆U?
Which of the following system freely allows the exchange of energy and matter with its environment?
Define heat.
An ideal gas is compressed at a constant temperature. Its internal energy will ____________.
Two samples A and B, of a gas at the same initial temperature and pressure are compressed from volume V to V/2; A isothermally and B adiabatically. The final pressure of A will be ______.
Two cylinders A and B of equal capacity are connected to each other via a stopcock. A contains a gas at standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. The stopcock is suddenly opened. Answer the following:
What is the change in the temperature of the gas?
In thermodynamics, heat and work are ______.
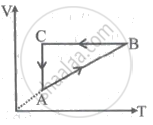
A cyclic process ABCA is shown in the V-T diagram. A process on the P-V diagram is ______.
The internal energy of one mole of argon is ______.
A system releases 125 kJ of heat while 104 kJ work is done on the system. Calculate the change in internal energy.
