Advertisements
Advertisements
Question
Complete the following table for homologous series of Alkenes.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Ethene | C2H4 | CH2 = CH2 | 2 | 0 | -102 |
| Propene | C3H6 | CH3–CH = CH2 | 3 | 1 | -48 |
| 1-Butene | C4H8 | CH3–CH2–CH = CH2 | ______ | ______ | -6.5 |
| 1-Pentene | C5H10 | CH3–CH2–CH2–CH = CH2 | ______ | ______ | 30 |
Advertisements
Solution
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Ethene | C2H4 | CH2 = CH2 | 2 | 0 | -102 |
| Propene | C3H6 | CH3–CH = CH2 | 3 | 1 | -48 |
| 1-Butene | C4H8 | CH3–CH2–CH = CH2 | 4 | 2 | -6.5 |
| 1-Pentene | C5H10 | CH3–CH2–CH2–CH = CH2 | 5 | 3 | 30 |
APPEARS IN
RELATED QUESTIONS
State any four characteristics of a homologous series
Write the name and formula of the 2nd member of homologous series having general formula CnH2n + 2.
Write the molecular formula of two consecutive members of homologous series of aldehydes. State which part of these compounds determines their
- physical and
- chemical properties
Write the molecular formula of the 2nd and 3rd member of the homologous series where the first member is ethyne.
Fill in the following blank with suitable word:
Ethene and ethyne are examples of ..... hydrocarbons.
Fill in the following blank with suitable word:
Carbon compounds have usually ... melting points and boiling points because they are ...... in nature.
The number of carbon atoms present in the molecule of fifth member of the homologous series of alkynes is:
(a) four
(b) five
(c) six
(d) seven
The molecular formula of the third member of the homologous series of ketones is:
(a) C4H8O
(b) C3H6O
(c) C5H10O
(d) C6H12O
Succeeding members of a homologous series differ by ______.
Give the names and the structural formula of the first three members of the homologous series of alkanes.
Write the name and molecular formula of the first member of the homologous series of alkynes.
Give the names of the first four members of the homologous series of
alkynes.
Copy and complete the following table, which relates to the three homologous series of hydrocarbons:
| General formula | CnH2n | CnH2n-2 | CnH2n+2 |
| IUPAC name of the homologus series | |||
| Characteristic bond type | Single bonds | ||
| IUPAC name of the first member of the series | |||
| Type of reaction with chlorine | Addition |
Observe the structural formula and answer the following questions.
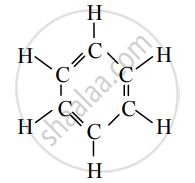
- Write the name of the given hydrocarbon.
- The given hydrocarbon is included in which type of hydrocarbon?
- What is the kind of compounds with the above characteristic structure called?
Which of the following does not belong to the same homologous series?
Define Homologous series.
Name and draw the electron dot structure of first homologue of alkynes series.
Name the third homologue of aldehydes.
