Advertisements
Advertisements
Question
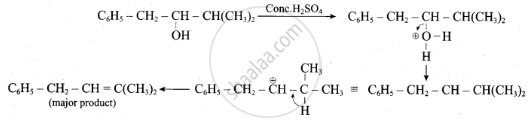
Complete the following reaction.
\[\ce{C6H5 - CH2CH(OH)CH(CH3)2 ->[conc. H2SO4]}\]
Advertisements
Solution
APPEARS IN
RELATED QUESTIONS
The reaction is an example of
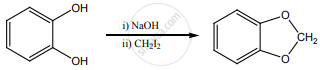
Arrange the following in the increasing order of their boiling point and give a reason for your ordering.
Propan-1-ol, propan-1, 2, 3-triol, propan-1, 3-diol, propan-2-ol
Write the chemical equation for Williamson's synthesis of 2-ethoxy-2-methyl pentane starting from ethanol and 2-methyl pentan-2-ol.
How is the conversion effected benzyl alcohol to benzoic acid?
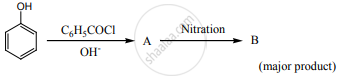
Complete the following reaction.
\[\begin{array}{cc}\phantom{..........}\ce{CH3}\\\phantom{.......}/\\\ce{C6H4}\\
\phantom{.......}\backslash\\\phantom{.........}\ce{OH}\end{array}\] is a/an ______.
Identify the product (s) is / are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
What will be the product (X and A) for the following reaction?
acetylchloride\[\ce{->[i) CH3MgBr][ii) H3O^+]X->[acid K2Cr2O7]A}\]
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
The correct IUPAC name of the compound,
\[\begin{array}{cc}
\ce{CH3}\phantom{......}\\
|\phantom{........}\\
\ce{H3C - CH - CH - CH - CH2 - OH}\\
|\phantom{............}|\phantom{........}\\
\ce{Cl}\phantom{...........}\ce{CH3}\phantom{......}
\end{array}\]
