Advertisements
Advertisements
प्रश्न
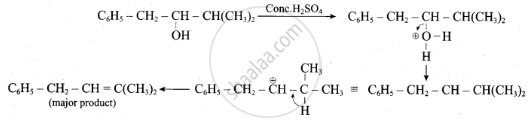
Complete the following reaction.
\[\ce{C6H5 - CH2CH(OH)CH(CH3)2 ->[conc. H2SO4]}\]
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Which of the following compounds on reaction with methyl magnesium bromide will give tertiary alcohol.
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Acid catalysed hydration
0.44 g of a monohydric alcohol, when added to methyl magnesium iodide in ether, liberates at STP 112 cm3 of methane with PCC the same alcohol form a carbonyl compound that answers the silver mirror test. Identify the compound.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
What will be the product (X and A) for the following reaction:
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O+] X ->[acid K2Cr2O7]A}\]
What will be the product (X and A) for the following reaction:
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O+]X->[acid K2Cr2O7]A}\]
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in the following method.
Acid catalysed hydration
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in each of the following methods.
Acid catalysed hydration
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
