Advertisements
Advertisements
प्रश्न
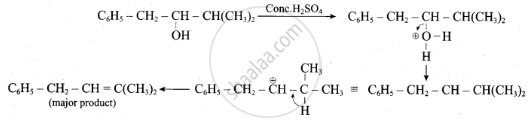
Complete the following reaction.
\[\ce{C6H5 - CH2CH(OH)CH(CH3)2 ->[conc. H2SO4]}\]
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Carbolic acid is ____________.
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Acid catalysed hydration
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroboration
Can we use nucleophiles such as NH3, CH3O for the Nucleophilic substitution of alcohols?
What will be the product (X and A) for the following reaction?
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+] X ->[acid K2Cr2O7] A}\]
\[\begin{array}{cc}\phantom{..........}\ce{CH3}\\\phantom{.......}/\\\ce{C6H4}\\
\phantom{.......}\backslash\\\phantom{.........}\ce{OH}\end{array}\] is a/an ______.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
What will be the product (X and A)for the following reaction
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O] X ->[acid K2Cr2O7] A}\]
What will be the product (X and A)for the following reaction
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+]X ->[acid K2Cr2O7]A}\]
