Advertisements
Advertisements
Question
Complete and balance the following equation:

Advertisements
Solution
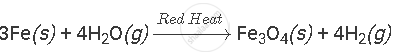
APPEARS IN
RELATED QUESTIONS
Write equations for the reactions of calcium and potassium with water.
Why are metals called electropositive elements whereas non-metals are called electronegative elements?
Complete and balance the following equation:
Na + O2 →
What happens when magnesium reacts with very dilute nitric acid? Write an equation for the reaction involved.
What happens when a rod of zinc metal is dipped into a solution of copper sulphate? Give chemical equation of the reaction involved.
A copper plate was dipped in AgNO3 solution. After certain time, silver from the solution was deposited on the copper plate. State the reason why it happened. Give the chemical equation of the reaction involved.
Write the equation for the reaction of Aluminium with dilute hydrochloric acid.
Name the products formed. Also indicate the physical states of all the substances involved.
Write the equation for the reaction of Iron with dilute hydrochloric acid.
Name the products formed. Also indicate the physical states of all the substances involved.
A basic oxide will be formed by the element:
(a) K
(b) S
(c) P
(d) Kr
Which one of the following four metals would be displaced from the solution of its salt by the other three metals?
(a) Zn
(b) Ag
(c) Cu
(d) Mg
Which of the following reactions will not occur? Why not?
(a) MgSO4 (aq) + Cu (s) → CuSO4 (aq) + Mg (s)
(b) CuSO4 (aq) + Fe (s) → FeSO4 (aq) + Cu (s)
(c) MgSO4 (aq) + Fe (s) → FeSO4 (aq) + Mg (s)
A zinc plate was kept in a glass container having CuSO4 solution. On examining it was found that the blue colour of the solution is getting lighter and lighter. After a few days, when the zinc plate was taken out of the solution, a number of small holes were noticed in it. State the reason and give chemical equation of the reaction involved.
What is the cause of chemical bonding (or chemical combination) of atoms of elements?
Choose the correct alternative and rewrite the following:
Reaction of iron nails whith copper sulphate solution is an example of _____________________
Choose the correct alternative and rewrite the following:
Which of the following is lue in colour?
Explain the following:
Reactivity of aluminium decrease if it is dipped in nitric acid.
Where does the metal aluminium, used in the thermit process, occurs in the reactivity series of metals?
Write the chemical equation for the following reaction:
Manganese dioxide is heated with aluminum powder.
An alkali metal A gives a compound B (molecular mass = 40) on reacting with water. The compound B gives a soluble compound C on treatment with aluminium oxide. Identify A, B and C and give the reaction involved.
The substance that will be flattened on beating with a hammer is ______.
