Advertisements
Advertisements
प्रश्न
Complete and balance the following equation:

Advertisements
उत्तर
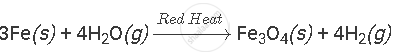
APPEARS IN
संबंधित प्रश्न
Write equations for the reactions of calcium and potassium with water.
Write the electron-dot structures for sodium, oxygen and magnesium.
Show the formation of Na2O by the transfer of electrons.
Differentiate between metal and non-metal on the basis of their chemical properties.
Which of the following elements would yield an acidic oxide.
Na, S, C, K, H
Which of the following elements would yield a basic oxide.
Na, S, C, K, H
What is the nature of the oxide Na2O? What happens when it is dissolved in water? Write the chemical equation of the reaction involved.
What happens when calcium reacts with chlorine? Write an equation for the reaction which takes place
Give one example, with equation, of the displacement of hydrogen by a metal from an acid.
You are given samples of three metals − sodium, magnesium and copper. Suggest any two activities to arrange them in order of their decreasing reactivities.
Write the chemical equation of the reaction which takes place when iron reacts with dilute sulphuric acid. What happens when the gas produced is ignited with a burning matchstick?
Write the equation for the reaction of Aluminium with dilute hydrochloric acid.
Name the products formed. Also indicate the physical states of all the substances involved.
Name one metal which displaces copper from copper sulphate solution and one which does not.
An element X reacts with hydrogen, when heated, to form a covalent hydride H2X. If H2X has a smell of rotten eggs, the element X is likely to be:
(a) carbon
(b) sulphur
(c) chlorine
(d) phosphorus
The elements whose oxides can turn litmus solution red are:
(a) lithium and sodium
(b) copper and potassium
(c) carbon and hydrogen
(d) phosphorus and sulphur
Which of the following pair of reactants can undergo a displacement reaction under appropriate conditions?
(a) MgSO4 + Fe
(b) ZnSO4 + Fe
(c) MgSO4 + Pb
(d) CuSO4 + Fe
CuSO4(aq) + Fe(s) → FeSO4(aq) + Cu(s)
FeSO4(aq) + Zn(s) → ZnSO4(aq) + Fe(s)
On the basis of the above reactions, indicate which is most reactive and which is least reactive metal out of zinc, copper and iron.
Explain the following:
Reactivity of aluminium decrease if it is dipped in nitric acid.
Metals are generally solid. Which of the following metals is in the liquid state at room temperature?
Find the odd one out:
