Advertisements
Advertisements
Question
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{Pb(NO3)2 + HCl ->}\]
Advertisements
Solution
\[\ce{Pb(NO3)2 + 2HCl -> PbCl2 ↓ + 2HNO3}\]
Dilute HCl is used.
APPEARS IN
RELATED QUESTIONS
Hydrochloric acid contains (i) hydrogen (ii) chlorine. Prove it. Write equations for the reactions.
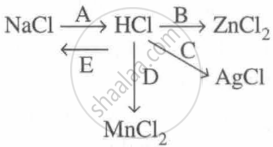
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
Name the following :
Acid present in the stomach of mammals.
Give a balanced equation when dilute hydrochloric acid is added to : Sodium sulphite
Give a balanced equation when dilute hydrochloric acid is added to : Magnesium bicarbonate
Give a balanced equation when dilute hydrochloric acid is added to : Potassium bisulphite
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl->}\]
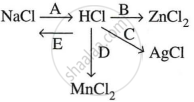
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.