Advertisements
Advertisements
Question
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NaHSO3 + HCl ->}\]
Advertisements
Solution
APPEARS IN
RELATED QUESTIONS
Name a chloride which is solube in excess of ammonium hydroxide
Give reason for the following:
silver nitrate solution can be used to distinguish HCl from HNO3
Name the following :
Acid present in the stomach of mammals.
Name the following:
Acid used to extract glue from bones.
Choose the correct answer from the options given below:
Which of the following statement is not correct ?
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl->}\]
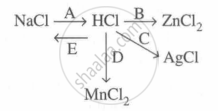
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
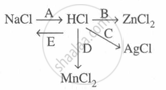
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCI->}\]
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
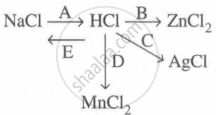
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.