Advertisements
Advertisements
प्रश्न
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NaHSO3 + HCl ->}\]
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Name the experiment which demonstrates that hydrogen chloride is soluble in water.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{Pb3O4 + HCl ->}\]
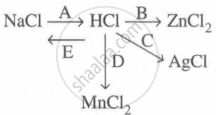
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
Name the following: A greenish yellow gas.
Give a balanced equation when dilute hydrochloric acid is added to : Zinc Metal
Choose the correct answer from the options given below:
Which of the following statement is not correct ?
Convert Hydrochloric acid to nascent chlorine.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
NH4OH + HCl ⟶
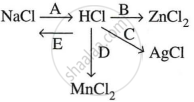
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.