Advertisements
Advertisements
Question
By drawing an electron dot diagram, show the lone pair effect leading to the formation of ammonium ion from ammonia gas and hydrogen ion.
Advertisements
Solution
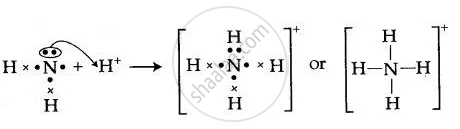
NH3 has one lone pair of electrons which is donates to hydrogen atom forming a co-ordinate bond. The arrow represents a co-ordinate bond. The arrow points from donor to acceptor atom.
APPEARS IN
RELATED QUESTIONS
Identify the term/substance in the following:
The electrons present in the outermost shell of an atom.
Identify the anion present in the following compounds:
A salt M on treatment with concentrated sulphuric acid produces a gas which fumes in moist air and gives dense fumes with ammonia
Choose the correct answer from the options given below:
A compound X consists of only molecules. Hence, X will have
A. A crystalline hard structure
B. A low melting point and low boiling point
C. An ionic bond
D. A strong force of attraction between its molecules
Give two example in the following case:
Co-ordinate bond compounds
What is meant by the term chemical bond and chemical bonding?
Fill in the blanks
A coordinate bond is represented by an arrow pointing from ________ to__________ atom.
Choose the correct answer from the options given below
Which one is coordinate molecule?
Draw an electron dot diagram of a hydronium ion and label the lone pair of electrons.
Bonding in this molecule can be understood to involve coordinate bonding.
What are the term defined below?
A bond formed by a shared pair of electrons with both electrons coming from the same atom.
