Advertisements
Advertisements
प्रश्न
By drawing an electron dot diagram, show the lone pair effect leading to the formation of ammonium ion from ammonia gas and hydrogen ion.
Advertisements
उत्तर
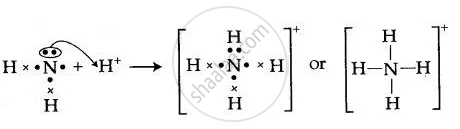
NH3 has one lone pair of electrons which is donates to hydrogen atom forming a co-ordinate bond. The arrow represents a co-ordinate bond. The arrow points from donor to acceptor atom.
APPEARS IN
संबंधित प्रश्न
Identify the term/substance in the following:
The electrons present in the outermost shell of an atom.
By drawing an electron dot diagram show the formation of Ammonium ion [Atomic No.: N = 7 and H = 1]
The following table shows the test a student performed on four different aqueous solutions which are X, Y, Z and W. Based on the observations provided, identify the cation present:
| Chemical test | Observation | Conclusion |
| To solution X, ammonium hydroxide is added in minimum quantity first and then in excess |
A dirty white precipitate is formed which dissolves in excess to form a clear solution |
(1) |
| To solution Y ammonium hydroxide is added in the minimum quantity first and then in excess |
A pale blue precipitate is formed which dissolves in excess to form a clear inky blue solution. |
(2) |
| To solution W a small quantity of sodium hydroxide solution is added and then in excess. |
A white precipitate is formed which remains insoluble. |
(3) |
| To a salt Z calcium hydroxide the solution is added and then heated. |
A pungent smelling gas turning moist red litmus paper blue is obtained. |
(4) |
Give a reason for Inert gases do not form ions.
Give the conditions for coordinate bond formation.
Fill in the blank
Coordinate bond is also called __________bond.
Fill in the blanks
A coordinate bond is represented by an arrow pointing from ________ to__________ atom.
Choose the correct answer from the options given below
Which one is coordinate molecule?
Give suitable chemical terms for the bond formed by a shared pair of electrons with both electrons coming from the same atom.
Bonding in this molecule can be understood to involve coordinate bonding.
