Advertisements
Advertisements
Question
Briefly explain the elementary particles present in nature.
Advertisements
Solution
- An atom has a nucleus surrounded by electrons and the nucleus is made up of protons and neutrons. Till 1960s, it was thought that protons, neutrons and electrons are fundamental building blocks of matter.
- In 1964, physicists Murray Gellman and George Zweig theoretically proposed that protons and neutrons are not fundamental particles; in fact they are made up of quarks. These quarks are now considered elementary particles of nature. Electrons are fundamental or elementary particles because they are not made up of anything.
- In the year 1968, the quarks were discovered experimentally by Stanford Linear Accelerator Centre (SLAC), USA. There are six quarks namely, up, down, charm, strange, top and bottom and their antiparticles. All these quarks have fractional charges.
For example, a charge of up quark is +`2/3`e and that of down quark is - `1/3`e. - According to the quark model, a proton is made up of two up quarks and one down quark, and a neutron is made up of one up quark and two down quarks.
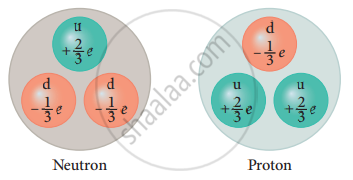
Constituents of nucleons - The study of elementary particles is called particle physics and it is an active area of research even now.
- Till date, more than 20 Nobel prizes have been awarded in the field of particle physics.
APPEARS IN
RELATED QUESTIONS
How long can an electric lamp of 100W be kept glowing by fusion of 2.0 kg of deuterium? Take the fusion reaction as
\[\ce{^2_1H + ^2_1H -> ^3_1He + n + 3.27 MeV}\]
Explain the processes of nuclear fission and nuclear fusion by using the plot of binding energy per nucleon (BE/A) versus the mass number A
Calculate the Q-values of the following fusion reactions :-
(a) `""_1^2H + ""_1^2H → ""_1^3H + ""_1^1H`
(b) `""_1^2H + ""_1^2H → ""_2^3H + n`
(c) `""_1^2H + ""_1^3H → _2^4H + n`.
Atomic masses are `m(""_1^2H) = 2.014102 "u", m(""_1^3H) = 3.016049 "u", m(""_2^3He) = 3.016029 "u", m(""_2^4He) = 4.002603 "u".`
(Use Mass of proton mp = 1.007276 u, Mass of `""_1^1"H"` atom = 1.007825 u, Mass of neutron mn = 1.008665 u, Mass of electron = 0.0005486 u ≈ 511 keV/c2,1 u = 931 MeV/c2.)
Why nuclear fusion reaction is also called thermo-nuclear reaction?
In our Nature, where is the nuclear fusion reaction taking place continuously?
A slab of stone of area 0.36 m2 and thickness 0.1 m is exposed on the lower surface to steam at 100°C. A block of ice at 0°C rests on the upper surface of the slab. In one hour 4.8 kg of ice is melted. The thermal conductivity of the slab is:
(Given latent heat of fusion of ice = 3.36 × 105 J kg−1)
The curve of binding energy per nucleon as a function of atomic mass number has a sharp peak for helium nucleus. This implies that helium nucleus is ______.
For a nuclear fusion process, the suitable nuclei are:
Solar energy is mainly caused due to:
How long can an electric lamp of 1000 W be kept glowing by fusion of 2.0 kg of deuterium? Take the fusion reaction as:
\[{}_{1}^{2}\mathrm{H}+{}_{1}^{2}\mathrm{H}\rightarrow{}_{2}^{3}\mathrm{He}+\mathrm{n}+3.27\mathrm{MeV}\]
