Advertisements
Advertisements
Question
Answer the following question.
Why are methane compounds have low melting and boiling points?
Advertisements
Solution
Covalent compounds have low intermolecular forces of attraction between the molecules and thus show low melting and boiling points. Since methane is also a covalent compound thus methane has very low melting and low boiling point.
RELATED QUESTIONS
What is the general name of all the compounds made up of carbon and hydrogen?
Choose the correct alternative.
Carbon forms innumerable compounds because
Name the following:
Substances whose atoms or molecules are not arranged in a geometrical pattern.
Carbon forms innumerable compounds by combining with other elements. True or false ?
Most of our food contains compounds of carbon. True or false ?
How is gas carbon produced?
Name of the following compounds :

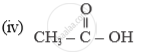
In which compound forms does carbon occur?
How would you prove experimentally that carbon dioxide does not support combustion.
How would you prove experimentally that is slightly acidic in nature.
Give a balanced equation for the following conversion: [In one or two steps]
A metallic oxide to calcium carbonate.
The property of self-linkage among identical atoms to form long chain compounds is known as ______.
Carbon exists in the atmosphere in the form of ______.
Explain the carbon cycle or biogeochemical cycle.
Carbon is one of the most important ______ elements.
Which of the following is highly toxic?
Carbon monoxide enters the human body through ______.
Answer the following by rearranging the jumbled letters :
The reaction of carbon with oxygen gives osdiex
Which theory was challenged by the synthetic preparation of Urea?
