Advertisements
Advertisements
प्रश्न
Answer the following question.
Why are methane compounds have low melting and boiling points?
Advertisements
उत्तर
Covalent compounds have low intermolecular forces of attraction between the molecules and thus show low melting and boiling points. Since methane is also a covalent compound thus methane has very low melting and low boiling point.
संबंधित प्रश्न
Fill in the blank.
The name ‘carbon’ is derived from the Latin word...........
Write true or false
Carbon constitues 0.03% of the earth’s crust.
Give two uses for the following:
sugar charcoal
What happens when a baking mixture containing baking powder is heated?
FILL IN THE BLANK
Organic compounds have ........ boiling and melting points.
Write the name and the chemical formula of the simplest alkene.
Name a gas which, though a compound of carbon, is not studied under organic chemistry.
Define catenation.
What is hydrocarbon? Give its one example.
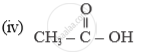
Name of the following compounds :

A carbon atom forms a ______ bond with other atoms. In this bond the two atoms ______ electrons.
(single, all, double, ionic, carbon, give and take, hydrogen, multiple, share, most, covalent)
Answer the following question.
Draw methane electron dot structure.
Answer the following question.
Name the type of bonds formed in the methane compound.
Answer the following question.
What happens when methane compound burns in oxygen?
How would you prove experimentally that is slightly acidic in nature.
Name the following:
An acid formed when carbon dioxide is dissolved in water under pressure.
Give a balanced equation for the following conversion: [In one or two steps]
Coke to water gas.
Structural formula of benzene is
Carbon monoxide enters the human body through ______.
How many valence electrons are there in carbon?
