Advertisements
Advertisements
Question
Answer the following question.
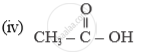
Draw methane electron dot structure.
Advertisements
Solution
Electron Dot structure of methane is:
RELATED QUESTIONS
Which of the following molecule is called buckministerfullerene?
C90 C60 C70 C120
Fill in the blank
................. is present in both living and non-living things.
Write true or false
Carbon constitues 0.03% of the earth’s crust.
Fill in the blank
...............is used in making black shoe polish.
Name the products formed when:
wood is burnt in the absence of air.
Give two uses for the following:
sugar charcoal
How will you collect the gas ?
Give reason for the following:
Soda acid and foam types of fire extinguisher are not used for extinguishing electrical fires.
State three ways by which carbon dioxide gas is added into the atmosphere.
Name of the following compounds :

In which compound forms does carbon occur?
A carbon atom forms a ______ bond with other atoms. In this bond the two atoms ______ electrons.
(single, all, double, ionic, carbon, give and take, hydrogen, multiple, share, most, covalent)
Answer the following question.
Name the type of bonds formed in the methane compound.
Answer the following question.
What happens when methane compound burns in oxygen?
Starting from carbon dioxide how would you obtain a black particles of carbon.
[Give balanced equations for the same]
State how you would convert carbon dioxide to a metallic carbonate using a basic oxide e.g. sodium oxide. [Give a balanced equation]
Select the appropeiate option of the following statement:
An allotrope of carbon obtained by burning kerosene oil in a limited supply of air.
Give a balanced equation for the following conversion: [In one or two steps]
A metallic oxide to calcium carbonate.
Carbon monoxide enters the human body through ______.
