Advertisements
Advertisements
Question
Answer the following question.
Name the type of bonds formed in the methane compound.
Advertisements
Solution
The type of bonds present in methane is all covalent bonds between four hydrogen atoms and the single carbon atom at the center of the molecule.
RELATED QUESTIONS
Name the scientist who disproved the 'vital force theory' for the formation of organic compounds.
Write true or false
Carbon constitues 0.03% of the earth’s crust.
Fill in the blank
...............is used in making black shoe polish.
Define the following:
Adsorption
Name the products formed when:
wood is burnt in the absence of air.
How will you collect the gas ?
What steps should be taken to balance carbon dioxide in the atmosphere ?
What is hydrocarbon? Give its one example.
Name of the following compounds :

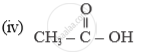
List the conclusions you will draw while studying the following properties of ethanoic acid :
(a) Odour
(b) Solubility in water
(c) Effect on litmus paper
(d) Reaction with sodium hydrogen carbonate
In which compound forms does carbon occur?
How would you prove experimentally that is slightly acidic in nature.
Explain the term ‘dry ice’. State its application.
Select the appropeiate option of the following statement:
The type of coal with the highest carbon conent.
Select the appropeiate option of the following statement:
An allotrope of carbon obtained by burning kerosene oil in a limited supply of air.
Answer the following by rearranging the jumbled letters :
Organic compounds having a double bond between carbon atoms are
knelaes
Which theory was challenged by the synthetic preparation of Urea?
