Advertisements
Advertisements
Question
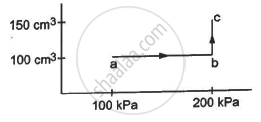
An ideal gas (γ = 1.67) is taken through the process abc shown in the figure. The temperature at point a is 300 K. Calculate (a) the temperatures at b and c (b) the work done in the process (c) the amount of heat supplied in the path ab and in the path bcand (d) the change in the internal energy of the gas in the process.
Advertisements
Solution
(a) For line ab, volume is constant.
So, from the ideal gas equation,
`"P"_1/"T"_1 = "P"_2/"T"_2`
`=> 100/300 = 200 /"T"_2`
`"T"_2 = ((200 xx 300 )/100) = 600 "K"`
For line bc, pressure is constant.
`So, "V"_1 /"T"_1 = "V"_2 /"T"_2`
`=> 100/600 = 150 / "T"_2`
`=> "T"_2 ((600 xx 150) /100) = 900 "K"`
(b)
As process ab is isochoric, Wab =0 .
During process bc,
P = 200 kPa
The volume is changing from 100 to 150 cm3 .
Therefore, work done = 50 × 10−6 × 200 × 103 J
= 10 J
(c) For ab (isochoric process), work done = 0.
From the first law,
dQ = dU = nCvdT
⇒ Heat supplied = nCvdT
Now,
`"Q"_"ab" = (("P""V")/("R""T")) xx (("R")/(gamma - 1)) xx "d""T"`
`= (200 xx 10 ^ 3 xx 100 xx 10 ^-6 xx 300)/(600 xx 0.67)`
= 14.925 ( ∴ γ = 1.67)
F or bc (isobaric process) :
Heat supplied in bc = `"n" "C"_"p""d""T" ( "C"_"p" = (gamma "R")/(gamma - 1))`
`= ("P""V")/("R""T") xx (gamma "R") /(gamma -1 ) xx "d""T"`
`= (200 xx 10^3 xx 150 xx 10 ^-6 )/(600 xx 0.67) xx 300 `
`= 10 xx 1.67 /0.67 = 16.7/0.67 = 24.925`
(d) dQ = dU + dW
Now,
dU = dQ − dW
= Heat supplied − Work done
= (24.925 + 14.925) − 10
= 39.850 − 10 = 29.850 J.
APPEARS IN
RELATED QUESTIONS
Which of the following quantities is zero on an average for the molecules of an ideal gas in equilibrium?
Keeping the number of moles, volume and temperature the same, which of the following are the same for all ideal gases?
Consider the quantity \[\frac{MkT}{pV}\] of an ideal gas where M is the mass of the gas. It depends on the
Calculate the volume of 1 mole of an ideal gas at STP.
Let Q and W denote the amount of heat given to an ideal gas and the work done by it in an isothermal process.
Let Q and W denote the amount of heat given to an ideal gas and the work done by it in an adiabatic process.
(a) Q = 0
(b) W = 0
(c) Q = W
(d) Q ≠ W
A rigid container of negligible heat capacity contains one mole of an ideal gas. The temperature of the gas increases by 1° C if 3.0 cal of heat is added to it. The gas may be
(a) helium
(b) argon
(c) oxygen
(d) carbon dioxide
A vessel containing one mole of a monatomic ideal gas (molecular weight = 20 g mol−1) is moving on a floor at a speed of 50 m s−1. The vessel is stopped suddenly. Assuming that the mechanical energy lost has gone into the internal energy of the gas, find the rise in its temperature.
An ideal gas is taken through a process in which the pressure and the volume are changed according to the equation p = kV. Show that the molar heat capacity of the gas for the process is given by `"C" ="C"_"v" +"R"/2.`
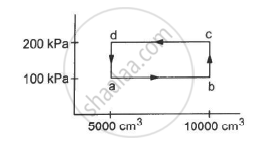
Half mole of an ideal gas (γ = 5/3) is taken through the cycle abcda, as shown in the figure. Take `"R" = 25/3"J""K"^-1 "mol"^-1 `. (a) Find the temperature of the gas in the states a, b, c and d. (b) Find the amount of heat supplied in the processes ab and bc. (c) Find the amount of heat liberated in the processes cd and da.
The volume of an ideal gas (γ = 1.5) is changed adiabatically from 4.00 litres to 3.00 litres. Find the ratio of (a) the final pressure to the initial pressure and (b) the final temperature to the initial temperature.
Figure shows a cylindrical tube with adiabatic walls and fitted with an adiabatic separator. The separator can be slid into the tube by an external mechanism. An ideal gas (γ = 1.5) is injected in the two sides at equal pressures and temperatures. The separator remains in equilibrium at the middle. It is now slid to a position where it divides the tube in the ratio 1 : 3. Find the ratio of the temperatures in the two parts of the vessel.

Two vessels A and B of equal volume V0 are connected by a narrow tube that can be closed by a valve. The vessels are fitted with pistons that can be moved to change the volumes. Initially, the valve is open and the vessels contain an ideal gas (Cp/Cv = γ) at atmospheric pressure p0 and atmospheric temperature T0. The walls of vessel A are diathermic and those of B are adiabatic. The valve is now closed and the pistons are slowly pulled out to increase the volumes of the vessels to double the original value. (a) Find the temperatures and pressures in the two vessels. (b) The valve is now opened for sufficient time so that the gases acquire a common temperature and pressure. Find the new values of the temperature and pressure.
ABCDEFGH is a hollow cube made of an insulator (Figure). Face ABCD has positive charge on it. Inside the cube, we have ionized hydrogen. The usual kinetic theory expression for pressure ______.

- will be valid.
- will not be valid since the ions would experience forces other than due to collisions with the walls.
- will not be valid since collisions with walls would not be elastic.
- will not be valid because isotropy is lost.
Diatomic molecules like hydrogen have energies due to both translational as well as rotational motion. From the equation in kinetic theory `pV = 2/3` E, E is ______.
- the total energy per unit volume.
- only the translational part of energy because rotational energy is very small compared to the translational energy.
- only the translational part of the energy because during collisions with the wall pressure relates to change in linear momentum.
- the translational part of the energy because rotational energies of molecules can be of either sign and its average over all the molecules is zero.
When an ideal gas is compressed adiabatically, its temperature rises: the molecules on the average have more kinetic energy than before. The kinetic energy increases ______.
- because of collisions with moving parts of the wall only.
- because of collisions with the entire wall.
- because the molecules gets accelerated in their motion inside the volume.
- because of redistribution of energy amongst the molecules.
We have 0.5 g of hydrogen gas in a cubic chamber of size 3 cm kept at NTP. The gas in the chamber is compressed keeping the temperature constant till a final pressure of 100 atm. Is one justified in assuming the ideal gas law, in the final state?
(Hydrogen molecules can be consider as spheres of radius 1 Å).
