Advertisements
Advertisements
Question
Accomplish the following conversion:
Aniline to p-bromoaniline
Advertisements
Solution
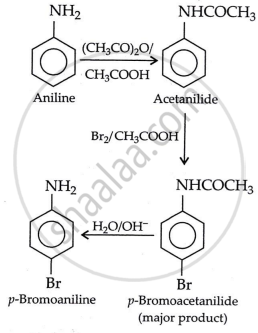
APPEARS IN
RELATED QUESTIONS
Arrange the following: C2H5NH2, C2H5OH, (CH3)3N – in the increasing order of their boiling point
- Write structures of different isomeric amines corresponding to the molecular formula C4H11N.
- Write the IUPAC names of all the isomers.
- What type of isomerism is exhibited by different pairs of amines?
How will you convert Benzene into aniline?
Arrange the following in increasing order of their basic strength:
C2H5NH2, C6H5NH2, NH3, C6H5CH2NH2 and (C2H5)2NH
Arrange the following in increasing order of their basic strength:
C2H5NH2, (C2H5)2NH, (C2H5)3N, C6H5NH2
Arrange the following in increasing order of their basic strength:
CH3NH2, (CH3)2NH, (CH3)3N, C6H5NH2, C6H5CH2NH2
Arrange the following:
In increasing order of boiling point:
C2H5OH, (CH3)2NH, C2H5NH2
Give reasons CH3NH2 is more basic than C6H5NH2.
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Choose the most correct option.
Which one of the following compounds has the highest boiling point?
Tertiary amines have lowest boiling points because ________________
Arrange the following compounds in increasing order of their boiling points.
Ethyl alcohol, Ethyl amine, Ethanoic acid, Ethane
The CORRECT decreasing order of solubility in water will be ____________.
Which among the following has the highest boiling point?
Which of the following should be most volatile?
Assertion: N-Ethylbenzene sulphonamide is soluble in alkali.
Reason: Hydrogen attached to nitrogen in sulphonamide is strongly acidic.
Which of the following amines form maximum hydrogen bonds within themselves?
