Advertisements
Advertisements
Question
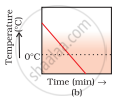
A glass tumbler containing hot water is kept in the freezer compartment of a refrigerator (temperature < 0°C). If you could measure the temperature of the content of the tumbler, which of the following graphs (Fig.1.2) would correctly represent the change in its temperature as a function of time.
Options
Advertisements
Solution
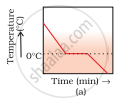
The hot water in the glass tumbler kept in the freezer will first become cold and the temperature will drop till 0°C. At 0°C, water loses heat equal to the latent heat of fusion till entire water freezes to form ice at 0°C. During this change of state from liquid to solid, the temperature remains constant.
On still further cooling, the temperature of ice slowly falls with time. Therefore, the correct option is (a).
APPEARS IN
RELATED QUESTIONS
The temperature at which a solid convert into a liquid is called its .........
Molecules of a substance are always in a state of............ and so they possess .............
Explain the term Melting.
Describe an experiment to demonstrate that a substance absorbs heat during melting without change in its temperature.
Some ice is taken in a beaker and its temperature is recorded after each one minute. The observations are listed below
| Time (in minute) | Temperature (in °C) |
| 0 | 0 |
| 1 | 0 |
| 2 | 0 |
| 3 | 0 |
| 4 | 0 |
| 5 | 0 |
| 6 | 3.8 |
| 7 | 7.6 |
| 8 | 11.4 |
From the above observations, what conclusion do you draw about the melting point of ice?
What are the changes of state in water? Explain.
What is melting? Give example.
A puddle of water gets pooled around the glass of ice cream or a glass of ice cubes. When it is kept at room temperature. Give reason.
Define latent heat of fusion?
Explain the following effects of heat.
Change in state