Advertisements
Advertisements
Rainbow is a beautiful natural phenomenon. It is the combined effect of a natural three processes together produced by light. Write it into the circle.


Concept: undefined >> undefined
Observe the given figure and write appropriate phenomenon of light in the box.

Concept: undefined >> undefined
Advertisements
Observe the given figure and answer the following questions.

- Which colour light rays bend most?
- Which colour light rays bend least.
- What is the wavelength of violet light rays?
Concept: undefined >> undefined
Observe the following diagram and identify the type of reaction and write observation.

Concept: undefined >> undefined
Explain why spacecraft take longer to reach the moon than light?
Concept: undefined >> undefined
______ colour is deviated the least in the spectrum of white light obtained with a glass prism.
Concept: undefined >> undefined
Observe the given figure of Fleming's Left Hand Rule and write the labels of 'A' and 'B':

Concept: undefined >> undefined
Identify the type of chemical reaction given below:
\[\ce{CuSO4 + Fe -> FeSO4 + Cu}\]
Concept: undefined >> undefined
Complete the following chemical reaction:
\[\ce{CuSO4 (aq) + Pb (s) -> \underline{}\underline{}\underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}\underline{}\underline{}}\]
Name the chemical reaction type.
Concept: undefined >> undefined
Metal ‘A’ has electronic configuration 2, 8, 1 and metal ‘B’ has electronic configuration 2, 8, 8, 2. Out of these, which metal is more reactive? Write the reaction of this metal with dilute HCl acid.
Concept: undefined >> undefined
Complete the given chemical equation:
\[\ce{Zn(s) + CuSO4(aq) -> \underline{}\underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}\underline{}}\]
Name the type of reaction.
Concept: undefined >> undefined
When a spacecraft is launched into outer space, the velocity attained by it should be greater than the escape velocity.
Concept: undefined >> undefined
Match the column:
| Column ‘A’ | Column ‘B’ |
| Alkali metals | (a) Group 18 |
| Halogens | (b) Group 1 |
| Noble gases | (c) Group 17 |
Concept: undefined >> undefined
Match the column:
| Column ‘A’ | Column ‘B’ |
| Alkali metals | (a) Group 18 |
| Halogens | (b) Group 1 |
| Noble gases | (c) Group 17 |
Concept: undefined >> undefined
Draw the ray diagram of a glass slab having medium A and B for the velocity of light ray VA and VB respectively and define Snell's law.
- If VB = 1.5 VA, then which medium is denser?
- What is the refractive index of A with respect to B?
- What is the refractive index of B with respect to A?
Concept: undefined >> undefined
State True or False:
"Wavelength of red light is close to 700 nm.”
Concept: undefined >> undefined
Observe the figure and name the ray AB, ray CD, ray GH.

Concept: undefined >> undefined
Observe the given diagram and answer the questions:
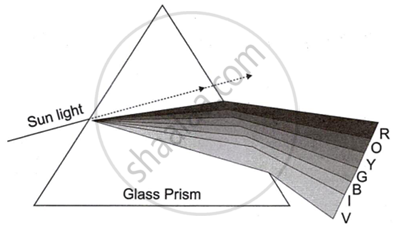
- Name the process shown in the figure.
- Name the colour that deviates the most.
- Name the colour that deviates the least.
- Name any one phenomenon in nature which is based on the above process.
- Define ‘spectrum’.
Concept: undefined >> undefined
In series combination which remains constant?
(a) Voltage
(b) Current
(c) Both current and voltage
(d) Both are variables
Concept: undefined >> undefined
Write two applications of sodium bicarbonate (baking soda).
Concept: undefined >> undefined
