Please select a subject first
Advertisements
Advertisements
State the law which is represented by the following graph:
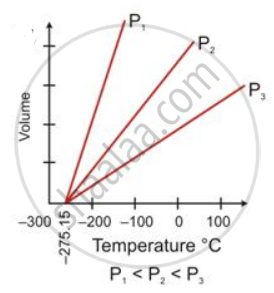
Concept: undefined >> undefined
Give reasons for the following:
Mountaineers carry oxygen cylinders with them.
Concept: undefined >> undefined
Advertisements
How did Charles's law lead to the concept of an absolute scale of temperature?
Concept: undefined >> undefined
State the following:
Ice point in absolute temperature
Concept: undefined >> undefined
Choose the correct answer:
The absolute temperature value that corresponds to 27°C is
Concept: undefined >> undefined
Choose the correct answer:
The volume-temperature relationship is given by
Concept: undefined >> undefined
What will be the minimum pressure required to compress 500 dm3 of air at 1 bar to 200 dm3 temperature remaining constant?
Concept: undefined >> undefined
2 liters of gas is enclosed in a vessel at a pressure of 760 mmHg. If the temperature remains constant, calculate pressure when volume changes to 4 dm3.
Concept: undefined >> undefined
88 cm3 of nitrogen is at a pressure of 770 mm mercury. If the pressure is raised to 880 mmHg, find by how much the volume will diminish, the temperature remaining constant.
Concept: undefined >> undefined
Calculate the final volume of a gas 'X' if the original pressure of the gas at STP is doubled and its temperature is increased three times.
Concept: undefined >> undefined
It is found that on heating a gas its volume increases by 50% and its pressure decreases to 60% of its original value. If the original temperature was −15°C, find the temperature to which it was heated.
Concept: undefined >> undefined
2500 cm3 of hydrogen is taken at STP. The pressure of this gas is further increased by two and a half times (temperature remaining constant). What volume will hydrogen occupy now?
Concept: undefined >> undefined
Taking the volume of hydrogen as calculated in Q.19, what change must be made in Kelvin (absolute) temperature to return the volume to 2500 cm3 (pressure remaining constant)?
Concept: undefined >> undefined
An LPG cylinder can withstand a pressure of 14.9 atmospheres. The pressure gauge of the cylinder indicates 12 atmospheres at 27°C. Because of a sudden fire in the building, the temperature rises. At what temperature will the cylinder explode?
Concept: undefined >> undefined
Give its
(i) graphical representation,
(ii) mathematical expression
(iii) significance
Concept: undefined >> undefined
Correct the following statement:
The volume of a fixed mass of a gas is directly proportional to its temperature, pressure remaining constant.
Concept: undefined >> undefined
Name the gas that.
Turns moist starch iodide paper blue black.
Concept: undefined >> undefined
A certain mass of gas occupied 850 ml at a pressure of 760 mm of Hg. On increasing the pressure it was found that the volume of the gas was 75% of its initial value. Assuming constant temperature, find the final pressure of the gas?
Concept: undefined >> undefined
At a given temperature the pressure of a gas reduces to 75% of its initial value and the volume increases by 40% of its initial value. Find this temperature if the initial temperature was -10°C.
Concept: undefined >> undefined
