Please select a subject first
Advertisements
Advertisements
An organic compound (A) (molecular formula C8H16O2) was hydrolysed with dilute sulphuric acid to give a carboxylic acid (B) and an alcohol (C). Oxidation of (C) with chromic acid produced (B). (C) on dehydration gives but-1-ene. Write equations for the reactions involved.
Concept: undefined >> undefined
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
Methyl benzoate
Concept: undefined >> undefined
Advertisements
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
m-Nitrobenzoic acid
Concept: undefined >> undefined
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
p-Nitrobenzoic acid
Concept: undefined >> undefined
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
Phenylacetic acid
Concept: undefined >> undefined
How will you bring about the following conversion in not more than two steps?
Benazaldehyde to α-Hydroxyphenylacetic acid
Concept: undefined >> undefined
Complete the synthesis by giving missing starting material, reagent or product.
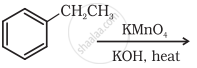
Concept: undefined >> undefined
Comment on the nature of two S−O bonds formed in SO2 molecule. Are the two S−O bonds in this molecule equal?
Concept: undefined >> undefined
How is the presence of SO2 detected?
Concept: undefined >> undefined
How is SO2 an air pollutant?
Concept: undefined >> undefined
Define the term Abnormal molar mass
Concept: undefined >> undefined
How will you convert the following in not more than two steps:
Benzoic acid to Benzaldehyde
Concept: undefined >> undefined
How will you convert the following in not more than two steps:
Acetophenone to Benzoic acid
Concept: undefined >> undefined
Write the coordination number and oxidation state of Platinum in the complex [Pt(en)2Cl2].
Concept: undefined >> undefined
Give reasons for the following
Elevation of the boiling point of 1 M KCl solution is nearly double than that of 1 M sugar solution.
Concept: undefined >> undefined
Give reason Aromatic diazonium salts are more stable than aliphatic diazonium salts.
Concept: undefined >> undefined
Name the reagents used in the following reactions:

Concept: undefined >> undefined
Account for the following:
PCl5 is more covalent than PCl3.
Concept: undefined >> undefined
Account for the following :
SnCl4 is more covalent than SnCl2.
Concept: undefined >> undefined
Predict whether van’t Hoff factor, (i) is less than one or greater than one in the following:
CH3COOH dissolved in water
Concept: undefined >> undefined
