Advertisements
Advertisements
Draw the structures of the following :
H2S2O7
Concept: undefined >> undefined
Which of the two components of starch is water soluble?
Concept: undefined >> undefined
Advertisements
Give one example each of sol and gel.
Concept: undefined >> undefined
Write equations of the following reactions:
Coupling reaction
Concept: undefined >> undefined
An unripe mango placed in a concentrated salt solution to prepare pickle, shrivels because ______.
Concept: undefined >> undefined
Consider the figure and mark the correct option.
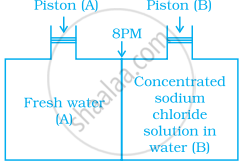
Concept: undefined >> undefined
Water retention or puffiness due to high salt intake occurs due to ______.
Concept: undefined >> undefined
Why are aquatic species more comfortable in cold water in comparison to warm water?
Concept: undefined >> undefined
When kept in water, raisin swells in size. Name and explain the phenomenon involved with the help of a diagram. Give three applications of the phenomenon.
Concept: undefined >> undefined
How will you distinguish between dispersed phase and dispersion medium in an emulsion?
Concept: undefined >> undefined
In the preparation of \[\ce{H2SO4}\] by Contact Process, why is \[\ce{SO3}\] not absorbed directly in water to form \[\ce{H2SO4}\]?
Concept: undefined >> undefined
Write the structure of pyrophosphoric acid.
Concept: undefined >> undefined
Match the formulas of oxides given in Column I with the type of oxide given in Column II and mark the correct option.
| Column I | Column II |
| (A) \[\ce{Pb3O4}\] | (1) Neutral oxide |
| (B) \[\ce{N2O}\] | (2) Acidic oxide |
| (C) \[\ce{Mn2O7}\] | (3) Basic oxide |
| (D) \[\ce{Bi2O3}\] | (4) Mixed oxide |
Concept: undefined >> undefined
Assertion: \[\ce{SF6}\] cannot be hydrolysed but \[\ce{SF4}\] can be.
Reason: Six \[\ce{F}\] atoms in \[\ce{SF6}\] prevent the attack of \[\ce{H2O}\] on sulphur atom of \[\ce{SF6}\].
Concept: undefined >> undefined
Which of the following compound will not undergo azo coupling reaction with benzene diazonium chloride?
Concept: undefined >> undefined
The product of the following reaction is:

(i)
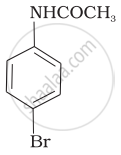
(ii)
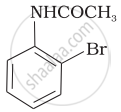
(iii)
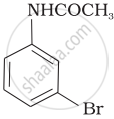
(iv)
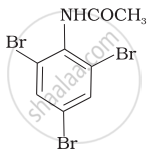
Concept: undefined >> undefined
Which of the following reactions belong to electrophilic aromatic substitution?
(i) Bromination of acetanilide
(ii) Coupling reaction of aryldiazonium salts
(iii) Diazotisation of aniline
(iv) Acylation of aniline
Concept: undefined >> undefined
Under what reaction conditions (acidic/basic), the coupling reaction of aryldiazonium chloride with aniline is carried out?
Concept: undefined >> undefined
Write the equations for the following reaction:
Salicylic acid is treated with acetic anhydride in the presence of conc H2SO4
Concept: undefined >> undefined
When benzene diazonium chloride reacts with phenol, it forms a dye. This reaction is called ______.
Concept: undefined >> undefined
