Advertisements
Advertisements
Cement, the important building material is a mixture of oxides of several elements. Besides calcium, iron and sulphur, oxides of elements of which of the group (s) are present in the mixture?
Concept: undefined >> undefined
Advertisements
Which of the following statements are correct. Answer on the basis of Figure.
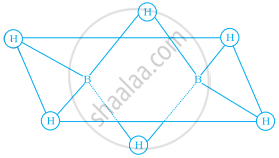
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iii) Out of six B – H bonds four B – H bonds can be described in terms of 3 centre 2 electron bonds;
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
Concept: undefined >> undefined
Explain why the following compounds behave as Lewis acids?
BCl3
Concept: undefined >> undefined
Explain why the following compounds behave as Lewis acids?
AlCl3
Concept: undefined >> undefined
When BCl3 is treated with water, it hydrolyses and forms [B[OH]4]– only whereas AlCl3 in acidified aqueous solution forms [Al(H2O)6]3+ ion. Explain what is the hybridisation of boron and aluminium in these species?
Concept: undefined >> undefined
Aluminium dissolves in mineral acids and aqueous alkalies and thus shows amphoteric character. A piece of aluminium foil is treated with dilute hydrochloric acid or dilute sodium hydroxide solution in a test tube and on bringing a burning matchstick near the mouth of the test tube, a pop sound indicates the evolution of hydrogen gas. The same activity when performed with concentrated nitric acid, reaction doesn’t proceed. Explain the reason.
Concept: undefined >> undefined
Explain the following:
Boron does not exist as B3+ ion.
Concept: undefined >> undefined
Explain the following:
PbX2 is more stable than PbX4.
Concept: undefined >> undefined
Explain the following:
Pb4+ acts as an oxidising agent but Sn2+ acts as a reducing agent.
Concept: undefined >> undefined
Explain the following:
Electron gain enthalpy of chlorine is more negative as compared to fluorine.
Concept: undefined >> undefined
Explain the following:
Tl (NO3)3 acts as an oxidising agent.
Concept: undefined >> undefined
Identify the compounds A, X and Z in the following reactions:
\[\ce{A + 2HCl + 5H2O -> 2NaCl + X}\]
Concept: undefined >> undefined
Identify the compounds A, X and Z in the following reactions:
\[\ce{X ->[Δ][370 K] HBO2 ->[Δ][> 370 K] Z}\]
Concept: undefined >> undefined
Complete the following chemical equations:
\[\ce{Z + 3 LiAlH4 -> X + 3LiF + 3AlF_3}\]
\[\ce{X + 6H2 -> Y + 6H2}\]
\[\ce{3X + 3O2 ->[Δ] B2O3 + 3H2O}\]
Concept: undefined >> undefined
Match the species given in Column I with the properties mentioned in Column II.
| Column I | Column II |
| (i) \[\ce{BF^{-}4}\] | (a) Oxidation state of central atom is +4 |
| (ii) AICI3 | (b) Strong oxidising agent |
| (iii) SnO | (c) Lewis acid |
| (iv) PbO2 | (d) Can be further oxidised |
| (e) Tetrahedral shape |
Concept: undefined >> undefined
Match the species given in Column I with properties given in Column II.
| Column I | Column II |
| (i) Diborane | (a) Used as a flux for soldering metals |
| (ii) Galluim | (b) Crystalline form of silica |
| (iii) Borax | (c) Banana bonds |
| (iv) Aluminosilicate | (d) Low melting, high boiling, useful for measuring high temperatures |
| (v) Quartz | (e) Used as catalyst in petrochemical industries |
Concept: undefined >> undefined
Match the species given in Column I with the hybridisation given in Column II.
| Column I | Column II |
| (i) Boron in [B(OH)4]– | (a) sp2 |
| (ii) Aluminium in [Al(H2O)6]3+ | (b) sp3 |
| (iii) Boron in B2H6 | (c) sp3d2 |
| (iv) Carbon in Buckminsterfullerene | |
| (v) Silicon in \[\ce{SiO^{4-}4}\] | |
| (vi) Germanium in [GeCl6]2– |
Concept: undefined >> undefined
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Atomic size
Concept: undefined >> undefined
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Ionisation enthalpy
Concept: undefined >> undefined
