Advertisements
Advertisements
प्रश्न
What are the common types of secondary structure of proteins?
Advertisements
उत्तर
The secondary structure of protein refers to the shape in which a long polypeptide chain can exist. They are found to exist in two different types of structures, viz., an α-helix and a β-pleated sheet structure. These structures arise due to the regular folding of the backbone of the polypeptide chain due to hydrogen bonding between \[\begin{array}{cc}\ce{O}\\||\\\ce{-C-}\end{array}\] and –NH– groups of the peptide bond.
α-Helix is one of the most common ways in which a polypeptide chain forms all possible hydrogen bonds by twisting into a right handed screw (helix) with the –NH group of each amino acid residue hydrogen bonded to the \[\begin{array}{cc}\backslash\phantom{.....}\\\ce{C=O}\\/\phantom{......}\end{array}\] of an adjacent turn of the helix as shown in the figure.

α-Helix structure of proteins
In β-pleated sheet structure, all peptide chains are stretched out to nearly maximum extension and then laid side by side, which are held together by intermolecular hydrogen bonds. The structure resembles the pleated folds of drapery and therefore is known as a β-pleated sheet.
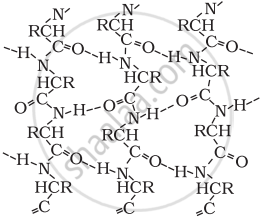
β-Pleated sheet structure of proteins
APPEARS IN
संबंधित प्रश्न
How are proteins classified on the basis of molecular shapes?
How is tripeptide formed?
What type of bonding helps in stabilising the α-helix structure of proteins?
The protein responsible for blood clotting is ____________.
The correct statement for protein haemoglobin.
Which of the following statement is correct:
α-Helix is a secondary structure of proteins formed by twisting of polypeptide chain into right-handed screw like structures. Which type of interactions are responsible for making the α-helix structure stable?
Each polypeptide in a protein has amino acids linked with each other in a specific sequence. This sequence of amino acids is said to be ______.
Peptide linkage is:
Explain formation of peptide linkage in protein with an example.
The total number of negative charge in the tetrapeptide, Gly-Glu-Asp-Tyr at pH 12.5 will be ______. (Integer answers)
Proteins are polymers of ______.
Assertion (A): Proteins are polymers of α-amino acids connected by a peptide bond.
Reason (R): A tetrapeptide contains 4 amino acids linked by 4 peptide bonds.
What is the effect of denaturation on the structure of proteins?
Statement I: A protein is imagined as a line, the left end represented by first amino acid (C-terminal) and the right end represented by last amino acid (Nterminal).
Statement II: Adult human haemoglobin, consists of 4 subunits (two subunits of a type and two subunits β type.)
In the light of the above statements, choose the correct answer from the options given below:
