Advertisements
Advertisements
प्रश्न
The variation of decay rate of two radioactive samples A and B with time is shown in figure.
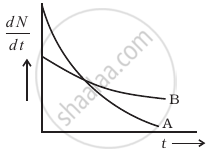
Which of the following statements are true?
- Decay constant of A is greater than that of B, hence A always decays faster than B.
- Decay constant of B is greater than that of A but its decay rate is always smaller than that of A.
- Decay constant of A is greater than that of B but it does not always decay faster than B.
- Decay constant of B is smaller than that of A but still its decay rate becomes equal to that of A at a later instant.
पर्याय
a and b
a and c
b and d
c and d
MCQ
Advertisements
उत्तर
c and d
Explanation:
It can be observed from the figure that the slope of curve A is greater than that of curve B, it means the rate of decay is faster for A than that of B.

According to Rutherford and Soddy's law for radioactive decay `-((dN)/(dt)) ∝ λ`, where decay. Hence at point P, the rate of decay for both A and B is the same.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
