Advertisements
Advertisements
प्रश्न
The beaker is divided into two chambers A and B. The big circle represents solute and the small circles solvent.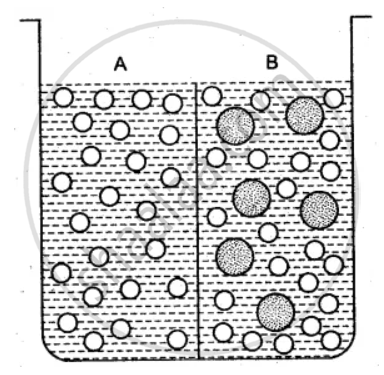
(i) What can you say about the size of the holes in the membrane, if it is to behave semi- permeably between these two?
(ii) Will the solvent molecules pass through the membrane from left to right, from right to left, in either direction or in both directions?
(iii) In which direction will there be a net movement of solvent molecules?
Advertisements
उत्तर
(i) The size of the holes in the membrane is large enough to allow only the solvent particles to pass through it. But solute particle cannot pass through it. Thus, the membrane acts as the semi-permeable.
(ii) Solvent molecules will pass through the membrane in both directions. Since solvent molecules are present on both the sides they will strike the semi-permeable membrane and pass through the same.
(iii) There is a net movement of solvent molecules from the place of its higher chemical potential to the place of its lower chemical potential, i.e., from right to left.
APPEARS IN
संबंधित प्रश्न
Give appropriate biological or technical terms for the following :
Movement of molecules of a substance from their higher concentration to lower concentration when they are in direct contact.
The bending movements of certain flowers towards the sun and the sleep movements of certain plants at night are examples of ______.
Give reason for the following:
Potato cubes, when placed in water, become firm and increase in size.
Leaves of the sensitive plant wilt and droop down on a slight touch. What mechanism brings about this change?
The diagram given below represents an experimental set-up to demonstrate a certain process. Study the same and answer the questions that follow:
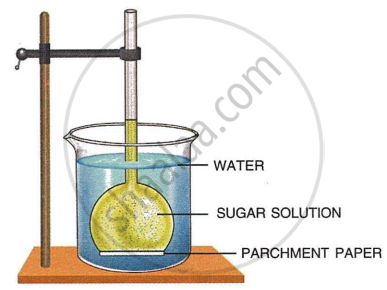
- Name the process.
- Define the above-named process.
- What would you observe in the experimental set-up after an hour or so?
- What control experiment can be set up for comparison?
- Keeping in mind the root hair, cell and its surroundings, name the parts that correspond to
- concentrated sugar solution
- parchment paper
- water in the beaker
- Name any other material that can be used instead of parchment paper in the above experiment.
- Mention two advantages of the process for the plants.
Two potato cubes each 1 cm3 in size, were placed separately in two containers (A&B), the container (A) having water and the other (B) containing concentrated sugar solution. After 24 hours when the cubes were examined, those placed in water were found to be firm and had increased slightly in size and those placed in concentrated sugar solution were found to be soft and had somewhat decreased in size. Use the above information to answer the questions that follow:
Account for the firmness and increase in the size of the potato cubes placed in water.
A leaf cell of a water plant was placed in a liquid other than pond water. After sometime, it assumed a shape as shown below:

Comment on the nature (tonicity) of the liquid surrounding the cell.
Fill in the Blank
The pressure which develops in the cortical cells of root which force a part of the water upward ____________.
Explain the Term: Exosmosis
Define the following term:
Osmotic Pressure
