Advertisements
Advertisements
प्रश्न
Define the following term:
Osmotic Pressure
Advertisements
उत्तर
Osmotic pressure is the minimum pressure that must be exerted to prevent the passage of the pure solvent into the solution when the two are separated by a semi-permeable membrane.
APPEARS IN
संबंधित प्रश्न
Osmosis and diffusion are the same except that osmosis there is:
The most appropriate characteristic of a semipermeable membrane is that
Give reason for the following:
Potato cubes, when placed in water, become firm and increase in size.
Give two examples of turgor movements in plants.
The diagram given below represents an experimental set-up to demonstrate a certain process. Study the same and answer the questions that follow:
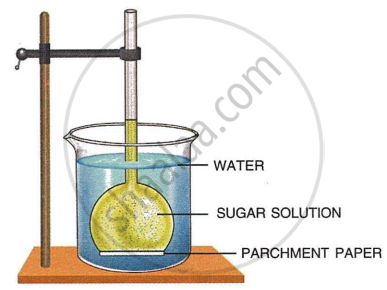
- Name the process.
- Define the above-named process.
- What would you observe in the experimental set-up after an hour or so?
- What control experiment can be set up for comparison?
- Keeping in mind the root hair, cell and its surroundings, name the parts that correspond to
- concentrated sugar solution
- parchment paper
- water in the beaker
- Name any other material that can be used instead of parchment paper in the above experiment.
- Mention two advantages of the process for the plants.
Two potato cubes each 1 cm3 in size, were placed separately in two containers (A&B), the container (A) having water and the other (B) containing concentrated sugar solution. After 24 hours when the cubes were examined, those placed in water were found to be firm and had increased slightly in size and those placed in concentrated sugar solution were found to be soft and had somewhat decreased in size. Use the above information to answer the questions that follow:
Account for the firmness and increase in the size of the potato cubes placed in water.
Define the following:
Osmosis
Name the following:
Movement of molecules from a region of high concentration to a region of low concentration.
Give Reasons for the following.
The raisins swell up in the water.
During a practical exam, a plant cell in a particular solution was placed under a compound microscope. Students were told to observe the cell and name the tonicity of the solution and mention the process that occurred in the cell.

