Advertisements
Advertisements
प्रश्न
Racemisation occurs in ______.
पर्याय
SN1 reaction
SN2 reaction
Neither SN1 nor SN2 reaction
SN2 reaction as well as SN1 reaction
Advertisements
उत्तर
Racemisation occurs in SN1 reaction.
Explanation:
A mixture containing two enantiomers in equal quantities will have zero optical rotation because the rotation caused by one isomer will be neutralised by the rotation caused by the other isomer. This type of mixture is known as a racemic mixture or racemic modification. Racemisation refers to the process of converting an enantiomer into a racemic mixture. Both retented and inverted products are generated during the SN1 reaction. Racemization happens as a result of the production of both d- and l- products.
APPEARS IN
संबंधित प्रश्न
Given reasons: C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
What are ambident nucleophiles? Explain with an example.
The stability order for carbocation is _______.
(A) 2° > 3° > 1°
(B) 3° > 2° > 1°
(C) 3° > 1° > 2°
(D) 1° > 3° > 2°
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
The reaction of C6H5–CH=CH–CH3 with HBr produces:
Which of the following statements are correct about the kinetics of this reaction?

(i) The rate of reaction depends on the concentration of only (b).
(ii) The rate of reaction depends on concentration of both (a) and (b).
(iii) Molecularity of reaction is one.
(iv) Molecularity of reaction is two.
When CH3CH2CHCl2 is treated NaNH2 product formed is:-
CCl4 is insoluble in water because:-
Give reason for the following:
The product formed during SN1 reaction is a racemic mixture.
The correct order of increasing reactivity of
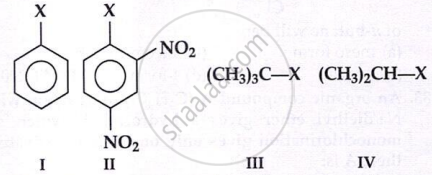
C-X bond towards nucleophile in the following compounds is:
