Advertisements
Advertisements
प्रश्न
Identify giving reason whether the following compound is aromatic or not.

Advertisements
उत्तर

The compound is non-aromatic since it has 4π electrons and hence, does not obey the Huckel rule of aromaticity.
APPEARS IN
संबंधित प्रश्न
Write the balanced chemical reaction to get benzene from Phenol.
Predict the possible product of the following reaction.
sulphonation of chlorobenzene
Predict the possible product of the following reaction.
nitration of toluene
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
Identify giving reason whether the following compound is aromatic or not.
Major product of the below mentioned reaction is, \[\ce{(CH3 )2 C = CH2 ->[ICI]}\]
Consider the nitration of benzene using mixed con H2SO4 and HNO3 if a large quantity of KHSO4 is added to the mixture, the rate of nitration will be ______.
Which of the following is aliphatic saturated hydrocarbon?
Give IUPAC name for the following compound.
\[\ce{CH ≡ C – C ≡ C – C ≡ CH}\]
Suggest the route for the preparation of the following from benzene.
4 – chlorotoluene
What happens when Isobutylene is treated with acidified potassium permanganate?
How will you prepare propane from a sodium salt of fatty acid?
Which of the following is NOT a hetero-aromatic compound?
Which among the following compounds has highest boiling point?
Phenol on distillation with zinc dust gives ____________.
Direct bromination of benzene with excess reagent results in the formation of ____________.
Which of the following molecules has shortest C - C bond length?
Which of the following compounds on bromination yields ![]() ?
?
The treatment of benzene with iso - butene in the presence of sulphuric acid gives:
Conversion of hexane into benzene involves the reaction of ______.
Arenes on treatments with chlorine in the presence of ferric chloride as a catalyst undergo what type of reaction?
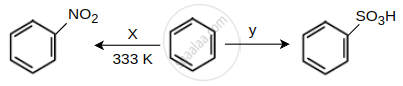
Identify the X and Y in the following reactions.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr->[Benzoyl][peroxide] H3C - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{.............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{...............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
