Advertisements
Advertisements
प्रश्न
Give one example of an endothermic reaction.
Advertisements
उत्तर
Example of endothermic reaction: Barium hydroxide on reacting with ammonium chloride produces ammonia, water and barium chloride. It also results in decrease in temperature, as it consumes heat energy.
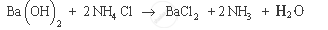
APPEARS IN
संबंधित प्रश्न
Why should a magnesium ribbon be cleaned before it is burnt in air?
2 mL of sodium hydroxide solution is added to a few pieces of granulated zinc metal taken in a test tube. When the contents are warmed, a gas evolves which is bubbled through a soap solution before testing. Write the equation of the chemical reaction involved and the test to detect the gas. Name the gas which will be evolved when the same metal reacts with dilute solution of a strong acid.
When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are elements.
With the help of an appropriate example, justify that some of the chemical reactions are determined by Change in temperature.
Give chemical equation for the reaction involved in the above case.
One of the following is an exothermic reaction. This is:
(a) electrolysis of water
(b) conversion of limestone into quicklime
(c) process of respiration
(d) process of photosynthesis
A metal X forms a salt XSO4. The salt XSO4 forms a clear solution in water which reacts with sodium hydroxide solution to form a blue precipitate Y. Metal X is used in making electric wire and alloys like brass.
(a) What do you think metal X could be?
(b) Write the name, formula and colour of salt XSO4.
(c) What is the blue precipitate Y?
(d) Write a chemical equation of the reaction which takes place when salt XSO4 reacts with sodium hydroxide solution. Give the state symbols of all the reactants and products which occur in the above equation.
Write word equation for the following skeletal equation:
\[\ce{FeCl2 + Cl2 -> FeCl3}\]
Write word equation for the following skeletal equation:
\[\ce{Ca + O2 → CaO}\]
In electrolysis of water, why is the volume of gas collected over one electrode double that of gas collected over the other electrode?
Balance the following equation:
Ca + N2 → Ca3N2
Balance the following equation:
Fe2O3 + CO → Fe + CO2
Balance the following equation:
Zn + HNO3 `→` Zn(NO3)2 + H2O + NO2
Write the balanced chemical equation of the following reaction.
barium chloride + sulphuric acid → barium sulphate + hydrochloric acid
Write the balanced chemical equation of the following reaction.
aluminium carbide + water → aluminium hydroxide + methane
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate the mass of salt formed.
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – zinc carbonate.
In certain reaction an insoluble solid called precipitate is formed. State the colour and name of the precipitate formed in the following reaction involving addition of:
Iron [III] chloride to ammonium hydroxide.
Balance the following simple equation:
KBr + Cl2 → KCl + Br2
In the chemical equation the _______ are written on the left-hand side.
The following reaction is an example of a `4"NH"_3("g") + "SO"_2 -> 4"NO"("g") + 6"H"_2"O"("g")`
- displacement reaction
- combination reaction
- redox reaction
- neutralisation reaction
