Advertisements
Advertisements
प्रश्न
Give one example of an endothermic reaction.
Advertisements
उत्तर
Example of endothermic reaction: Barium hydroxide on reacting with ammonium chloride produces ammonia, water and barium chloride. It also results in decrease in temperature, as it consumes heat energy.
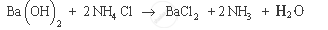
APPEARS IN
संबंधित प्रश्न
Why should a magnesium ribbon be cleaned before it is burnt in air?
Balance the following chemical equation.
\[\ce{BaCl2 + H2SO4 -> BaSO4 + HCl}\]
Write a balanced chemical equation for the process of photosynthesis giving the physical states of all the substances involved and the conditions of the reaction.
What happens when silver nitrate solution is added to sodium chloride solution?
Write the equation for the reaction which takes place.
Balance the following equation. Also name the product formed.
`"KCIO"_3 → "KCI" +"O"_2`
Write your observation and name the product when ron nails are added to an aqueous solution of copper sulphate.
Complete the following equation:
CH4 + O2 —>
Write the balanced chemical equation of the following reaction.
sodium chloride + manganese dioxide + sulphuric acid → sodium hydrogen sulphate + manganese sulphate + water + chlorine.
(a) Give only one suitable chemical test to identify the following gases.
- Ammonia
- Sulphur dioxide
- Hydrogen Chloride
- Chlorine
- Carbon Dioxide
- Oxygen
- Hydrogen
(b) Select a basic gas mentioned in Q.1 (a). How is the basic nature suspected?
(c)Select acidic gases from the gases mentioned in Q.1 (a). How is the acidic nature suspected?
(d) State the gas responsible for bleaching action.
(e)Which gas turn blue cobalt chloride paper light pink?
How is a flame test performed?
Define: Endothermic reaction
Write word equation for the following molecular equation:
AgN03 + HCI → HNO3 + AgCl ↓
Word equation:
State whether AgCl is soluble or insoluble in HN03.
Give word equation for the following chemical reaction and give the names of the product formed.
Zn + 2HC1→ ZnCl2 + H2
Name the following:
The gas evolved when a dilute acid is added to chalk [limestone].
Balance the following simple equation:
\[\ce{Ca(OH)2 + HNO3 -> Ca(NO3)2 + H2O}\]
Balance the following simple equation:
K + H2O → KOH + H2
Balance the following simple equation:
Mg + CO2 → MgO + C
CaCO3 + 2HCl[dil.] → CaCl2 + H2O + CO2 [g]
State the information not conveyed by the above chemical equation.
Give a balanced equation by partial equation method, [steps are given below].
The reaction of excess ammonia with chlorine – Ammonia as a reducing agent
- Ammonia first reacts with chlorine to give hydrogen chloride and nitrogen.
- Hydrogen chloride then further reacts with excess ammonia to give ammonium chloride.
To balance the following chemical equation the value of x and y should respectively be:
\[\ce{2NaOH + xAl_2O_3->yNaAlO_2 + H_2O}\]
