Advertisements
Advertisements
प्रश्न
Give a simple chemical test to distinguish between the following pair of compounds:
Phenol and Benzoic acid
Advertisements
उत्तर १
Phenol and benzoic acid can be distinguished by ferric chloride test. In the ferric chloride test, phenol reacts with neutral FeCl3 to make an iron-phenol complex that turns violet.
\[\ce{\underset{Phenol}{6C6H5OH} + FeCl3 -> \underset{(Violet colour)}{\underset{Iron-phenol complex}{[Fe(OC6H5)6]^3-}} + 3H+ + 3Cl-}\]
But benzoic acid reacts with neutral FeCl3 to give a buff coloured ppt. of ferric benzoate.
\[\ce{\underset{Benzoic acid}{3C6H5OH} + FeCl3 ->\underset{(Buff coloured ppt)}{\underset{Ferric benzoate}{(C6H5COO)3Fe}} + 3HCl}\]
उत्तर २
Benzoic acid reacts with NaHCO3 to release CO2 gas with effervescence, whereas phenol does not.
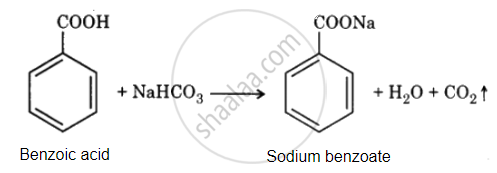
Phenol Br2 decolourises water to give white precipitate but does not give benzoic acid.

APPEARS IN
संबंधित प्रश्न
Out of CH3CH2 – CO – CH2 – CH3 and CH3CH2 – CH2 – CO – CH3, which gives iodoform test?
Give a simple chemical test to distinguish between the following pair of compounds:
Acetophenone and Benzophenone
Give a simple chemical test to distinguish between the following pair of compounds:
Benzoic acid and Ethyl benzoate
Give a simple chemical test to distinguish between the following pair of compounds:
Pentan-2-one and Pentan-3-one
Give a simple chemical test to distinguish between the following pair of compounds:
Benzaldehyde and Acetophenone
Alkenes decolourise bromine water in presence of CCl4 due to formation of ______.
Oxidation of ketones involves carbon-carbon bond cleavage. Name the products formed on oxidation of 2, 5-dimethylhexan-3-one.
Which sugar does not reduce Fehling's solution?
Which of the following tests/reactions is given by aldehydes as well as ketones?
A hydrocarbon (A) with molecular formula C5H10 on ozonolysis gives two products (B) and (C). Both (B) and (C) give a yellow precipitate when heated with iodine in presence of NaOH while only (B) give a silver mirror on reaction with Tollen’s reagent.
- Identify (A), (B) and (C).
- Write the reaction of B with Tollen’s reagent.
- Write the equation for iodoform test for C.
- Write down the equation for aldol condensation reaction of B and C.
The major products formed in the following reaction sequence A and B are:

An organic compound neither reacts with neutral ferric chloride solution nor with Fehling solution. It however, reacts with Grignard reagent and gives positive iodoform test. The compound is:
Choose the reaction which is not possible:
The reagent that can be used to distinguish acetophenone and benzophenone is ______.
An organic compound 'A' with molecular formula C5H8O2 is reduced to n-pentane with hydrazine followed by heating with NaOH and glycol. 'A' forms a dioxime with hydroxylamine and gives a positive iodoform and Tollen's test. Identify 'A' and give its reaction for iodoform and Tollen's test.
You are given four organic compounds “A”, “B” , “C” and “D”. The compounds “A”, “B” and “C” form an orange-red precipitate with 2, 4 DNP reagent. Compounds “A” and “B” reduce Tollen’s reagent while compounds “C” and “D” do not. Both “B” and “C” give a yellow precipitate when heated with iodine in the presence of NaOH. Compound “D” gives brisk effervescence with sodium bicarbonate solution. Identify “A”, “B”, “C” and “D” given the number of carbon atoms in three of these carbon compounds is three while one has two carbon atoms. Give an explanation for our answer.
Benzaldehyde is obtained from Rosenmund’s reduction of:
Match List-I with List-II:
| List-I (Reaction) |
List-II (Reagents/Condition) |
||
| A. | 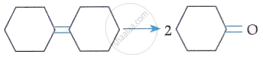 |
I. | 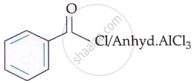 |
| B. | 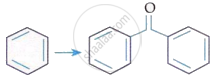 |
II. | CrO3 |
| C. | 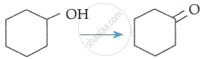 |
III. | KMnO4/KOH, Δ |
| D. | 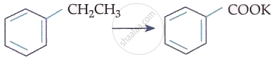 |
IV. | (i) O3 (ii) Zn-H2O |
Choose the correct answer from the options given below:
