Advertisements
Advertisements
प्रश्न
Find the kinetic energy of the emitted electron, if in photoelectric effect energy of incident Photon is 4 eV and work function is 2.4 eV.
Advertisements
उत्तर
K.E.max = hν − φ0
= 4 − 2.4
= 1.6 eV.
APPEARS IN
संबंधित प्रश्न
Explain the inverse linear dependence of stopping potential on the incident wavelength in a photoelectric effect experiment.
The electrons are emitted in the photoelectric effect from a metal surface.
As the intensity of incident light increases ______
Find the energy of photon which have momentum 2 × 10-16 gm-cm/sec.
Explain the concept of the photoelectric effect.
With the help of a circuit diagram describing an experiment to study the photoelectric effect.
What is the photoelectric effect? Define stopping potential and photoelectric work function.
The ratio of energies of photons produced due to transition of electron of hydrogen atom from its (i) second to first energy level and (ii) highest energy level to second level is respectively.
The graph of stopping potential `"V"_"s"` against frequency v of incident radiation is plotted for two different metals P and Q as shown in the graph. ΦP and ΦQ are work-functions of P and Q respectively, then
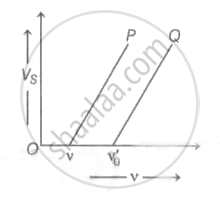
The maximum velocity of the photoelectron emitted by the metal surface is 'v '. Charge and mass of the photoelectron is denoted by 'e' and 'm' respectively. The stopping potential in volt is ______.
When a light of wavelength 4000 Å falls on a photoelectric emitter, photoelectrons are liberated. For another emitter, light of wavelength 6000 Å is sufficient for photo emission. The work functions of the two emitters are in the ratio of ____________.
Threshold frequency for a metal is 1015 Hz. Light of `lambda` = 4000 Å falls on its surface. Which of the following statements is correct?
A metal surface is illuminated by light of given intensity and frequency to cause photoemission. If the intensity of illumination is reduced to one-fourth of its original value then the maximum KE of the emitted photoelectrons would be ______.
The work function of a metallic surface is 5.01 eV. The photoelectrons are emitted when light of wavelength 2000 Å falls on it. The potential difference applied to stop the fastest photoelectrons is [h = 4.14 x 10-15 eV sec] ____________.
A metal surface is illuminated by photons of energy 5 eV and 2.5 eV respectively. The ratio of their wavelengths is ____________.
In photoelectric effect, graph of saturation current versus frequency of light is plotted. The nature of the graph will be ____________.
When wavelength of incident radiation on the metal surface is reduced from 'λ1' to 'λ2', the kinetic energy of emitted photoelectrons is tripled. The work function of the metal is ______.
(h = Planck's constant, c =velocity of light)
Following graphs show the variation of stopping potential corresponding to the frequency of incident radiation (F) for a given metal. The correct variation is shown in graph (v0 = Threshold frequency).
The work function of a photosensitive material is 4.0 eV. The longest wavelength of light that can cause photon emission from the substance is (approximately) ____________.
Which one of the following statements ts INCORRECT for stopping potential in photoelectric emission?
The radiations of energies 1 eV and 2.5 eV are incident on a metal surface having work function 0.5 eV. The ratio of the maximum velocities of the emitted photo-electrons is ____________.
Two incident radiations having energies two times and ten times of the work function of a metal surface, produce photoelectric effect. The ratio of maximum velocities of emitted photo electrons respectively is ____________.
The stopping potential in the context of photoelectric effect depends on the following property of incident electromagnetic radiation ______.
When ultraviolet light of wavelength 100 nm is incident upon a sample of silver metal, a potential difference of 7.7 volt is required to stop the photoelectrons from reaching the collector plate. The potential required to stop photo electrons when light of wavelength 200 nm is incident upon silver is ______.
In a photocell, frequency of incident radiation is increased by keeping other factors constant (v > v0), the stopping potential ______.
Light of wavelength λ, which is less than threshold wavelength is incident on a photosensitive material. If incident wavelength is decreased so that emitted photoelectrons are moving with same velocity, then stopping potential will ______.
Photoelectric emission is observed from a metallic surface for frequencies ν1 and ν2 of the incident light rays (ν1 > ν2). If the ratio of the maximum value of the kinetic energy of the photoelectrons emitted in the first case to that in the second case is 2 : K, then the threshold frequency of the metallic surface is ______.
The photoelectric threshold for a certain metal surface is 3600 Å. If the metal surface is irradiated by a wavelength of 1100 Å, then kinetic energy of the emitted photoelectrons is ______.
In a photoelectric experiment, the stopping potential is 1.5V. What is the maximum kinetic energy of a photoelectron?
