Advertisements
Advertisements
प्रश्न
The energy of a photon is 2 eV. Find its frequency and wavelength.
Advertisements
उत्तर
Given:
E = 2 eV = 2 × 1.6 × 10−19 = 3.2 × 10−19 J
To find:
- Frequency (ν)
- Wavelength (λ)
Formulae:
- E = hν
- λ = `c/ν`
Calculation:
i. Using formula (i),
v = `(3.2 xx 10^-19)/(6.63 xx 10^-34)`
= `(3.2 xx 10^15)/6.63`
= antilog {log (3.2) + log(1015) − log(6.63)}
= antilog {0.5051 + 15 − 0.8215}
= antilog {14.6836}
= 4.826 × 1014 Hz
The frequency of photons is 4.826 × 1014 Hz.
ii. Using formula (ii),
`λ = c/ν = (3 xx 10^8)/(4.826 xx 10^14)`
= antilog {log 3 − log 4.826} × 10−6
= antilog {0.4771 − 0.6836} × 10−6
= antilog {`overline1`.7935} × 10−6
= 0.6216 × 10−6m
= 6216 × 10−5 m
= 6216 Å
The wavelength of the photon is 6216 Å.
APPEARS IN
संबंधित प्रश्न
Can microwaves be used in the experiment on photoelectric effect?
Is it always possible to see the photoelectric effect with a red light?
Using the values of work function given in the following table, tell which metal will require the highest frequency of incident radiation to generate photocurrent.
Typical values of work function for some common metals
| Metal | Work function (in eV) |
| Potassium | 2.3 |
| Sodium | 2.4 |
| Calcium | 2.9 |
| Zinc | 3.6 |
| Silver | 4.3 |
| Aluminium | 4.3 |
| Tungsten | 4.5 |
| Copper | 4.7 |
| Nickel | 5.0 |
| Gold | 5.1 |
Radiation of wavelength 4500 Å is incident on a metal having work function 2.0 eV. Due to the presence of a magnetic field B, the most energetic photoelectrons emitted in a direction perpendicular to the field move along a circular path of radius 20 cm. What is the value of the magnetic field B?
As the intensity of incident light increases ______
The minimum frequency for photoelectric effect on metal is 7 × 1014 Hz, Find the work function of the metal.
The kinetic energy of the most energetic photoelectron emitted from a metal surface is doubled when the wavelength of the incident radiation is reduced from λ1 to λ2. The work function of the metal is ______
The following graph shows the stopping potential V0 versus frequency v for photoelectric emission from two metals A and B. The slope of each of the lines gives ______

The graph of stopping potential `"V"_"s"` against frequency v of incident radiation is plotted for two different metals P and Q as shown in the graph. ΦP and ΦQ are work-functions of P and Q respectively, then
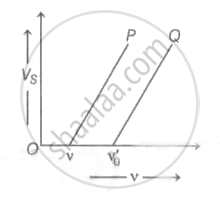
The maximum velocity of the photoelectron emitted by the metal surface is 'v '. Charge and mass of the photoelectron is denoted by 'e' and 'm' respectively. The stopping potential in volt is ______.
A metal surface is illuminated by light of given intensity and frequency to cause photoemission. If the intensity of illumination is reduced to one-fourth of its original value then the maximum KE of the emitted photoelectrons would be ______.
An important spectral emission line has a wavelength of 21 cm. The corresponding photon energy is (h = 6.62 x 10-34 Js, c = 3 x 108 m/s) ____________.
Threshold wavelength for lithium metal is 6250 Å. For photoemission, the wavelength of the incident light must be ______.
When certain metal surface is illuminated with a light of wavelength A., the stopping potential is V, When the same surface is illuminated by light of wavelength 2λ, the stopping potential is `("V"/3)`. The threshold wavelength for the surface is ______.
In photoelectric effect, graph of saturation current versus frequency of light is plotted. The nature of the graph will be ____________.
Light of frequency 2 times the threshold frequency is incident on a photo sensitive material. If the frequency is made `1/3`rd and intensity is doubled then the photocurrent will ______.
When wavelength of incident radiation on the metal surface is reduced from 'λ1' to 'λ2', the kinetic energy of emitted photoelectrons is tripled. The work function of the metal is ______.
(h = Planck's constant, c =velocity of light)
Photoelectrons are emitted from a photosensitive surface for the light of wavelengths λ1 = 360 nm and λ2 = 600 nm. What is the ratio of work functions for lights of wavelength 'λ1' to 'λ2'?
When a metal with work function 0.6 eV is illuminated with light of energy 2 eV, the stopping potential will be ____________.
When a photosensitive surface is irradiated by lights of wavelengths `lambda_1` and `lambda_2`, kinetic energies of emitted photoelectrons are E1 and E2 respectively. The work function of the photosensitive surface is ____________.
When the work function of a metal increases, maximum kinetic energy of emitted photoelectrons ____________.
Is it always necessary to use red light to get a photoelectric effect?
The wavelength of light incident on a metal surface is reduced from 300 nm to 200 nm (both are less than threshold wavelength). What is the change in the stopping potential for photoelectrons emitted from the surface will be ______ V. (Take h = 6.6 × 10-34 J-s)
For a given photosensitive material and frequency (> threshold frequency) of incident radiation, the photoelectric current varies with the intensity of incident light as:
On a photosensitive material when frequency of incident radiation is increased by 30%, kinetic energy of emitted photoelectrons increases from 0.4 eV. The work function of the surface is ______.
A parallel plate capacitor with air between the plates has capacitance 9 pF. The separation between the plates becomes thrice and the space between them is filled with a medium of dielectric constant 6. The capacitance becomes 'x' pF. The value of 'x' is ______.
