Advertisements
Advertisements
प्रश्न
Draw the TP diagram (P-x axis, T-y axis), VT(T-x axis, V-y axis) diagram for
- Isochoric process
- Isothermal process
- Isobaric process
Advertisements
उत्तर
a. Isobaric process: PV0 = nRT

P(T) = `("nRT")/"V"_0` and T(V) = Multivalued
b. Isothermal process: PV = nRT0

P(T) = Multivalued and T(V) = T0
c. Isobaric process: P0V = nRT
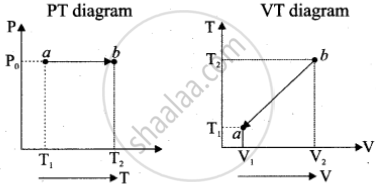
P(T) = P0 and T(V) = `("P"_0"V")/"nR"`
APPEARS IN
संबंधित प्रश्न
What is a thermodynamic process?
Draw a p-V diagram of the irreversible process.
The V-T diagram of an ideal gas which goes through a reversible cycle A→B→C→D is shown below. (Processes D→A and B→C are adiabatic)

The corresponding PV diagram for the process is (all figures are schematic)
Apply first law for an adiabatic process.
Apply first law for an isobaric process.
Draw the PV diagram for the adiabatic process.
Draw the PV diagram for the isochoric process.
Derive the work done in an adiabatic process.
Explain the isobaric process and derive the work done in this process.
In an adiabatic expansion of the air, the volume is increased by 4%, what is the percentage change in pressure? (For air γ = 1.4)
