Advertisements
Advertisements
प्रश्न
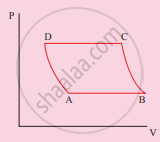
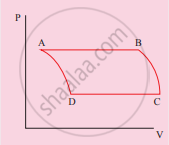
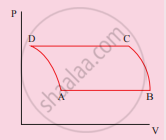
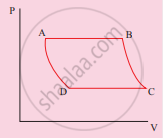
The V-T diagram of an ideal gas which goes through a reversible cycle A→B→C→D is shown below. (Processes D→A and B→C are adiabatic)

The corresponding PV diagram for the process is (all figures are schematic)
पर्याय
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Answer in brief.
Why should a Carnot cycle have two isothermal two adiabatic processes?
An ideal gas of volume 2 L is adiabatically compressed to (1/10)th of its initial volume. Its initial pressure is 1.01 x 105 Pa, calculate the final pressure. (Given 𝛾 = 1.4)
Write a note on free expansion.
Explain work done during a thermodynamic process.
Give an expression for work done in an isothermal process.
Apply first law for an isothermal process.
Give an equation state for an isochoric process.
Explain in detail the isochoric process.
For a given ideal gas 6 × 105 J heat energy is supplied and the volume of gas is increased from 4 m3 to 6 m3 at atmospheric pressure. Calculate
- the work done by the gas
- change in internal energy of the gas
- graph this process in PV and TV diagram
The work done on the system in changing the state of a gas adiabatically from equilibrium state A to equilibrium state B is 22.4 J. If the gas is taken from state A to B through another process in which the net heat absorbed by the system is 15.5 cal, then the net work done by the system in the latter case is ______.
( l cal = 4.2 J)