Advertisements
Advertisements
प्रश्न
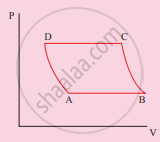
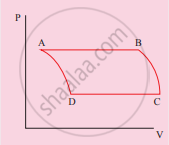
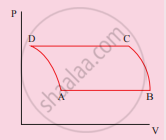
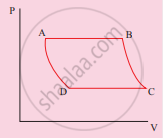
The V-T diagram of an ideal gas which goes through a reversible cycle A→B→C→D is shown below. (Processes D→A and B→C are adiabatic)

The corresponding PV diagram for the process is (all figures are schematic)
विकल्प
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Explain why The climate of a harbour town is more temperate than that of a town in a desert at the same latitude.
What is a thermodynamic process?
Draw a p-V diagram of the reversible process.
An ideal gas of volume 2 L is adiabatically compressed to (1/10)th of its initial volume. Its initial pressure is 1.01 x 105 Pa, calculate the final pressure. (Given 𝛾 = 1.4)
Explain in detail the isothermal process.
Explain in detail an adiabatic process.
Two identical samples of a gas are allowed to expand (i) isothermally (ii) adiabatically. Work done is ____________.
In which of the following processes, beat is neither absorbed nor released by a system?
In an isothermal process, the volume of an ideal gas is halved. One can say that ____________.
Explain how can a gas be expanded at constant temperature.