Advertisements
Advertisements
प्रश्न
Define: Atomic number
Advertisements
उत्तर
Atomic number— Atomic number refers to the number of protons present in an atom. It is denoted by Z.
APPEARS IN
संबंधित प्रश्न
Write down the name of a compound represented by the following formula:
K2SO4
Write down the name of a compound represented by the following formula:
CaCl2
Give the name of the element present in the following compound:
Quick lime
Give the name of the element present in the following compound:
Hydrogen bromide
Give the name of the element present in the following compound:
Baking powder
What is the atomicity of the following ?
- Oxygen
- Ozone
- Neon
- Sulphur
- Phosphorus
- Sodium
Write the formulae of the following compounds. Also name the elements present in them.
- Water
- Ammonia
- Methane
- Sulphur dioxide
- Ethanol
In what form does oxygen gas occur in nature ?
Work out the formula for magnesium hydrogencarbonate.
The cation of an element has :
An element A forms an oxide A2O5.
What is the valency of element A ?
An element X forms the following compounds with hydrogen, carbon and oxygen :
H2X, CX2, XO2, XO3
State the three valencies of element X which are illustrated by these compounds.
Explain the term – molecules. Give three examples of atoms of the same element forming a molecule. State the atomicity of the same.
Give one example of a triatomic molecule
Give a reason why –
Ozone is considered triatomic, but phosphorus a tetratomic molecule.
State the atomicity of the following:
Neon
State the atomicity of the following:
Argon
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 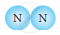 |
|
| 4. | 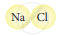 |
If a molecule is made of similar kinds of atoms, then it is called ______ atomic molecule.
The Atomicity of phosphorous is ______
Give any two examples for heterodiatomic molecules.
Which of the following contains maximum number of molecules?
Write any two classifications of a molecule.
