Advertisements
Advertisements
प्रश्न
Acetone, Acetaldehyde, Benzaldehyde, Acetophenone – reactivity towards addition of HCN.
Advertisements
उत्तर
Acetone, acetaldehyde, benzaldehyde, acetophenone - Reactivity with HCN
\[\begin{array}{c}\ce{CH3-C-CH3}\\\ce{||}\\\ce{O}\end{array}
\begin{array}{c}\ce{CH3-CHO}\\\ce{||}\\\ce{O}\end{array}\ce{C6H5CHO}\begin{array}{c}\ce{CH3-C-C6H5}\\\ce{||}\\\ce{O}\end{array}\]
Nucelophile addition by HCN is strongest for aldehyde than ketones. Also, acetophenone is more sterically hindered than acetone, hence comparatively less nucleophilic similarly C6 H5CHO i -s less reactive towards nucleophilic addition than CH3CHO.
\[\begin{array}{c}\ce{CH3-C-C6H5}\\\ce{||}\\\ce{O}\end{array}
\begin{array}{c}\ce{< CH3-C -CH3}\\\ce{||}\\\ce{O}\end{array}\ce{<C6H5CHO < CH3CHO}\]
APPEARS IN
संबंधित प्रश्न
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
H2N − NHCONH2/H+
How will you bring about the following conversion in not more than two steps?
Bromobenzene to 1-Phenylethanol
Complete the synthesis by giving missing starting material, reagent or product.
\[\ce{C6H5CHO ->[H2NCONHNH2]}\]
Write balanced chemical equations for action of ammonia on - acetone
A Idol condensation will not be observed in
Acetaldehyde and acetone differ in their reaction with
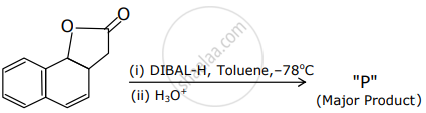
The product "P" in the above reaction is:
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
