Advertisements
Advertisements
Explain the following term with suitable examples:
Frenkel defect
Concept: undefined >> undefined
Explain the following term with a suitable example:
Interstitials
Concept: undefined >> undefined
Advertisements
Boiling point of water at 750 mm Hg is 99.63°C. How much sucrose is to be added to 500 g of water such that it boils at 100°C? Molal elevation constant for water is 0.52 K kg mol−1.
Concept: undefined >> undefined
Write the chemistry of recharging the lead storage battery, highlighting all the materials that are involved during recharging.
Concept: undefined >> undefined
Explain the term with suitable examples:
Alcosol
Concept: undefined >> undefined
Explain the term with suitable examples:
Aerosol
Concept: undefined >> undefined
Explain the term with suitable examples:
Hydrosol
Concept: undefined >> undefined
Aqueous copper sulphate solution (blue in colour) gives a green precipitate with aqueous potassium fluoride. Explain this experimental result.
Concept: undefined >> undefined
A solution of [Ni(H2O)6]2+ is green but a solution of [Ni(CN)4]2− is colourless. Explain.
Concept: undefined >> undefined
[Fe(CN)6]4− and [Fe(H2O)6]2+ are of different colours in dilute solutions. Why?
Concept: undefined >> undefined
What will be the correct order for the wavelengths of absorption in the visible region for the following?
[Ni(NO2)6]4−, [Ni(NH3)6]2+, [Ni(H2O)6]2+
Concept: undefined >> undefined
How do you explain the amphoteric behaviour of amino acids?
Concept: undefined >> undefined
Describe a method for the identification of primary, secondary and tertiary amines. Also write chemical equations of the reactions involved.
Concept: undefined >> undefined
Give a plausible explanation for the following:
Why do primary amines have higher boiling points than tertiary amines?
Concept: undefined >> undefined
Predict the product of the following reaction:
\[\ce{CH3 - CH2 - CH2 - O - CH3 + HBr ->}\]
Concept: undefined >> undefined
Predict the product of the following reaction:
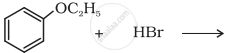
Concept: undefined >> undefined
Predict the product of the following reaction:
\[\ce{(CH3)3C - OC2H5 ->[HI]}\]
Concept: undefined >> undefined
Show how will you synthesize cyclohexylmethanol using an alkyl halide by an SN2 reaction.
Concept: undefined >> undefined
Write the equation of the reaction of hydrogen iodide with 1-propoxypropane.
Concept: undefined >> undefined
Write the equation of the reaction of hydrogen iodide with methoxybenzene.
Concept: undefined >> undefined
