Definitions [8]
Nomenclature is the system of assignment of names to organic compounds.
Compounds having the same molecular formula but different structural formula are known as Isomers and the phenomenon is known as Isomerism. (iso = same, meros = parts).
The stabilising interaction that involves delocalisation of σ-electrons of C–H bond of an alkyl group linked directly to an atom of unsaturated system or to an atom having unshared p-orbital, is called hyperconjugation.
e.g.
The change of a solid substance directly into a gas or vapour without first changing into a liquid is called sublimation.
OR
The change from solid state to vapour state without passing through the liquid state is called sublimation, and the substance is said to sublime.
Define sublimation.
The change of state from solid to gas directly is called sublimation.
Define Distillation
The process in which liquid is converted into its vapor phase at its boiling point and the vapor is then condensed back to liquid on cooling is known as distillation.
Define :- Distillation
Distillation : Distillation is the method of getting a pure liquid from a solution by evaporating and then condensing the vapours.
Define the Stationary phase.
Stationary phase is a solid or a liquid supported on a solid which remains fixed in a place and on which different solutes are adsorbed to a different extent.
Key Points
Carbon has 4 electrons in its outermost shell → forms 4 covalent bonds → tetravalent.
Determination of Hybridisation (First Method):
- Count the number of pi (π) bonds on that carbon atom.
- Single-bonded C → sp³ hybridisation
- C with one π bond → sp²
- C with two π bonds → sp
| Bonds on C | Hybridisation | Geometry | Example |
|---|---|---|---|
| 4 sigma, 0 pi | sp³ | Tetrahedral | Methane (CH₄) |
| 3 sigma, 1 pi | sp² | Trigonal planar | Ethene (C₂H₄) |
| 2 sigma, 2 pi | sp | Linear | Ethyne (C₂H₂) |
Shapes of molecules:
- Methane (CH₄) → sp³ hybrid orbitals
- Ethene (C₂H₄) → sp² hybrid orbitals
- Ethyne (C₂H₂) → sp hybrid orbitals
(i) Based on Functional Group:
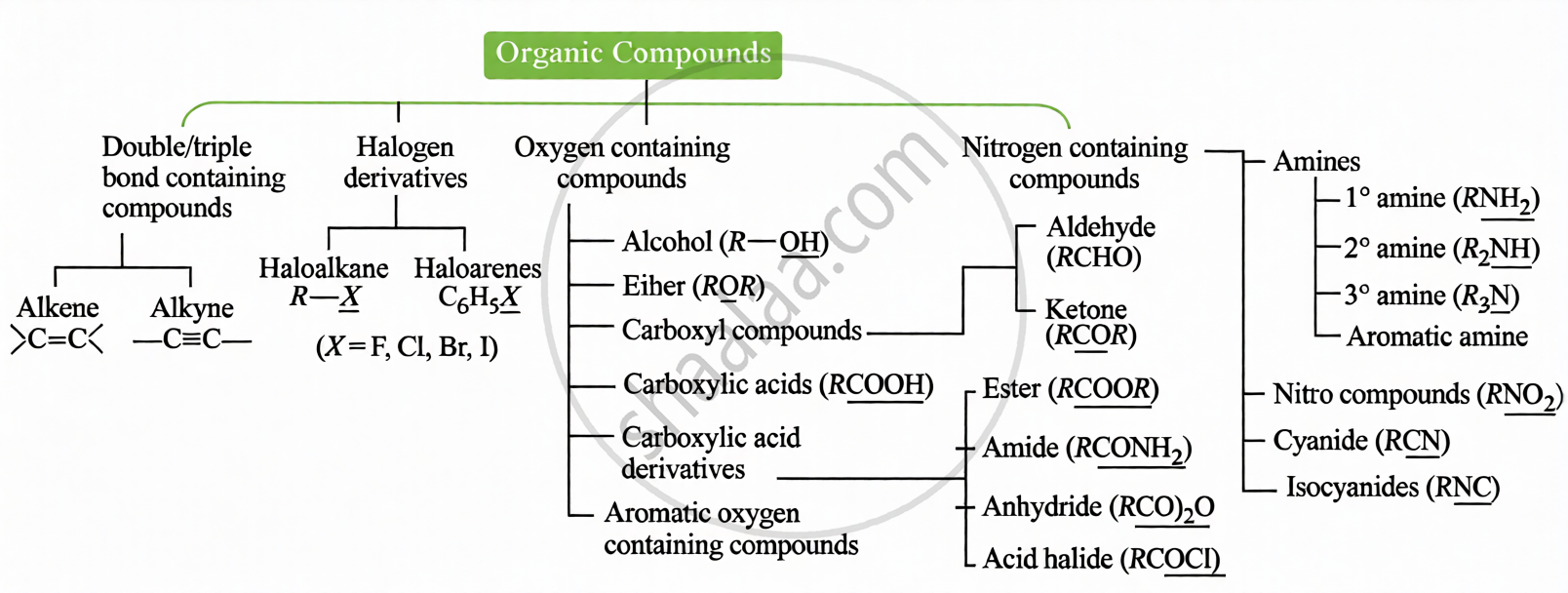
(ii) Based on Carbon Skeleton:
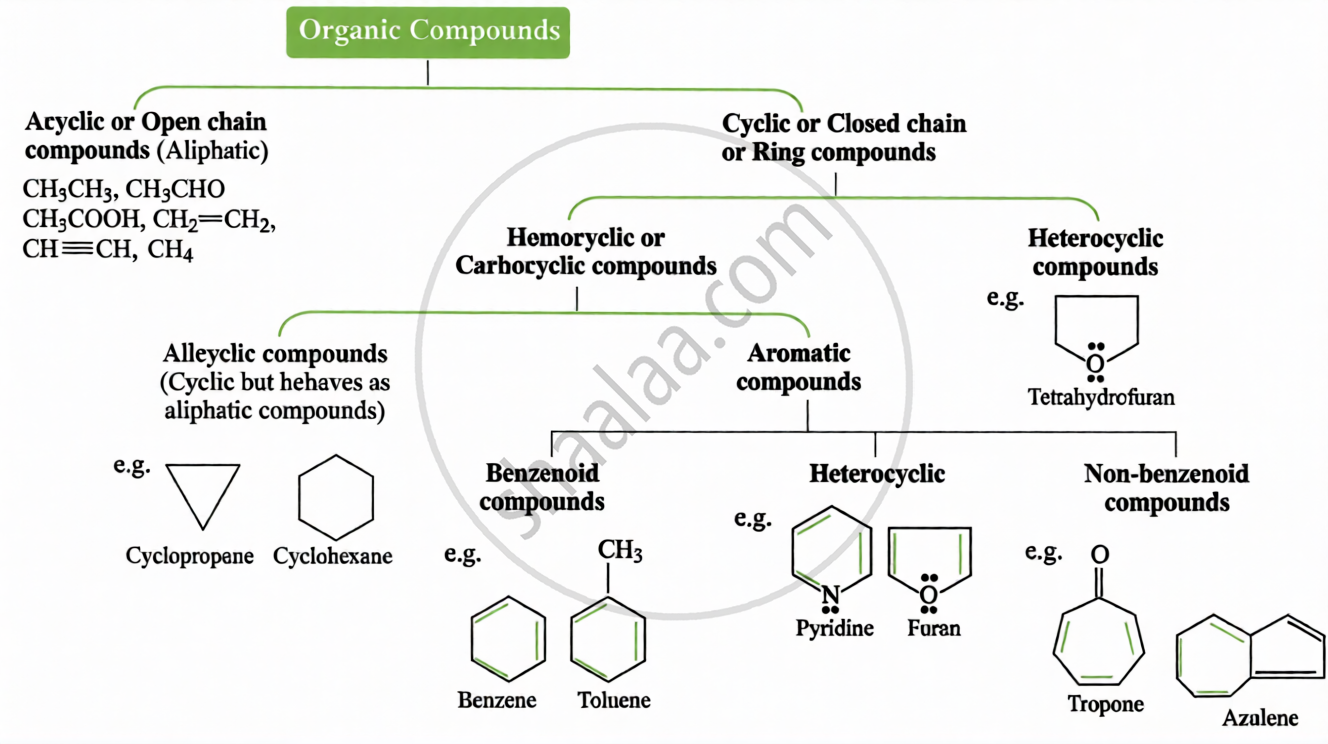
- The IUPAC system provides a unique, systematic way to name carbon compounds based on structure, replacing confusing common names.
- An IUPAC name has three parts: prefix, parent, and suffix, reflecting the carbon chain and functional group.
- The parent name is based on the longest carbon chain, and its ending changes to –ane, –ene, or –yne depending on the number of bonds.
- Functional groups are shown as prefixes or suffixes, and the chain is numbered to give them the lowest possible number.
- If the suffix begins with a vowel, the final ‘e’ in the parent alkane name is dropped (e.g., propane → propanone).
1. Structural Isomerism
Same molecular formula but different connectivity (bonding) of atoms.
Types:
- Chain isomerism: Different carbon skeleton (straight/branched)
- Position isomerism: Functional group at different positions
- Functional isomerism: Different functional groups (e.g., alcohol vs ether)
- Metamerism: Different alkyl groups on either side of same functional group
- Tautomerism: Dynamic equilibrium between two forms (keto ↔ enol)
2. Stereoisomerism
Same molecular formula and bonding but different spatial arrangement.
Types:
I. Geometrical isomerism:
- Due to restricted rotation (double bond)
- Forms cis (same side) and trans (opposite side)
II. Optical isomerism:
- Mirror image isomers (enantiomers)
- Show optical activity (rotate plane polarized light)
- Permanent effect due to shift of σ-electrons
- Electrons move towards more electronegative atom
- Effect decreases with distance (negligible after 3 carbons)
e.g-
![]()
| Type | Meaning | Example |
|---|---|---|
| –I effect | Electron withdrawing group | –NO₂, –Cl |
| +I effect | Electron donating group | –CH₃ |
Resonance:
- When a single Lewis structure cannot explain all properties of a compound, two or more structures (canonical forms) are written
- The real molecule does not exist as separate forms, it exists as a resonance hybrid
- Resonance hybrid is a weighted average of all canonical forms
- Due to resonance, electrons are delocalised over the molecule
- Resonance hybrid is more stable (lower energy) than any single structure
Resonance Effect (Mesomeric Effect):
| Type | Explanation | Effect on Electron Density | Examples |
|---|---|---|---|
| +R effect | Group donates electrons to conjugated system | Increases electron density at certain positions | –OH, –OR, –NH₂, –NHR |
| –R effect | Group withdraws electrons from conjugated system | Decreases electron density at o- and p-positions | –NO₂, –CN, –CHO, –COOH |
A temporary effect involving the complete transfer of shared π-electrons to one of the atoms in a multiple bond, in the presence of an attacking reagent.
| Type | Direction of π-electron transfer | Example |
|---|---|---|
| +E Effect | π electrons transferred to the atom to which the reagent gets attached | |
| –E Effect | π electrons transferred to the atom to which the reagent does not attach | 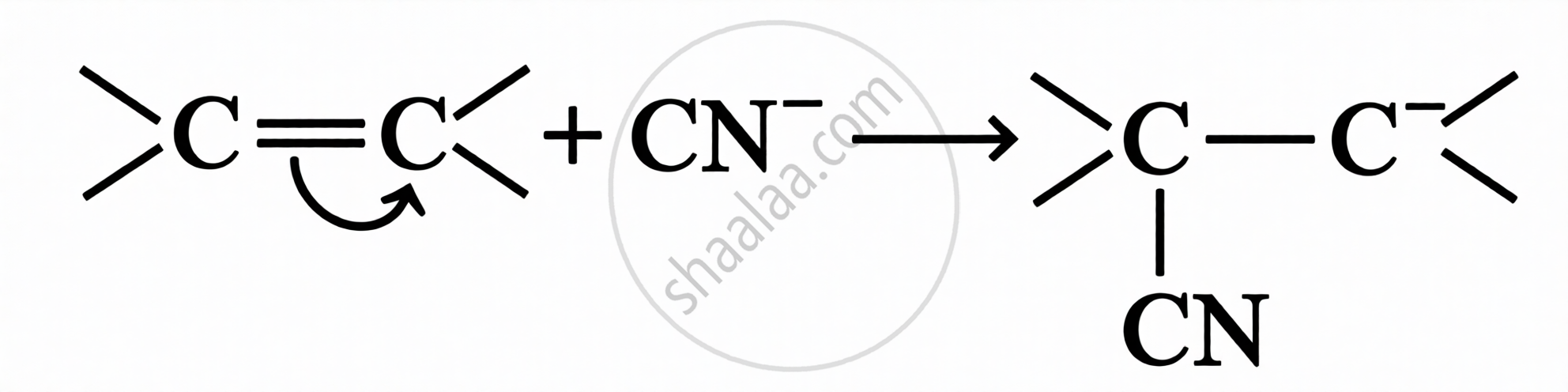 |
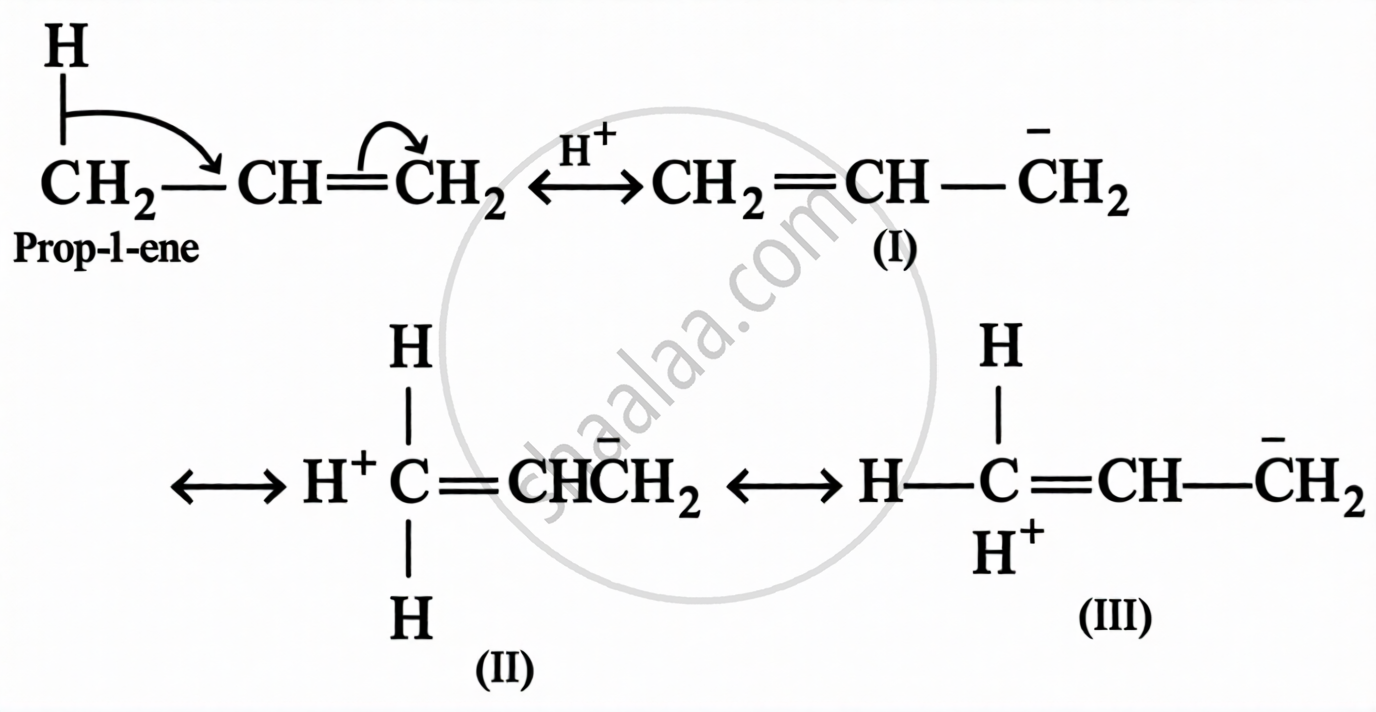
Also called the Baker-Nathan effect or no-bond resonance.
Conditions for hyperconjugation:
- The compound must have at least one sp²-hybridised carbon (alkene, alkyl carbocation, or alkyl free radical)
- The α-carbon must have at least one H–C bond attached to the unsaturated system
- More the number of H–C bonds at α-carbon → greater the stabilisation → more stable the alkene/carbocation
Resonance vs. Hyperconjugation:
| Feature | Resonance | Hyperconjugation |
|---|---|---|
| Electrons involved | π electrons delocalised | σ electrons of C–H bond delocalised |
| Stability provided | Greater | Lesser |
| Bond type | π bond | σ bond |
- Organic compounds obtained from natural sources or prepared in the laboratory are usually impure.
- Purification is required to obtain compounds in a pure form for the study of physical and chemical properties.
- Impurities may be solids, liquids, or gases mixed with the compound.
- Different purification methods are used based on physical properties like solubility, boiling point, and volatility.
- The choice of method depends on the nature of the compound and impurity.
- Based on difference in solubility of the compound and impurities at different temperatures.
- Compound more soluble in hot solvent → dissolved at high temperature → cooled → pure crystals form → impurities remain in solution (mother liquor).
- Common solvents: water, alcohol, ether, chloroform, acetone, benzene, petroleum ether.
- Example: Benzoic acid (slightly soluble in cold water, completely soluble in hot water) → separated from naphthalene.
- Fractional crystallisation: Used when two compounds have different solubilities in the same solvent → less soluble crystallises first on cooling (e.g., K₂CO₃ and KCl).
- Introduced by Tswett in 1906.
- Based on the difference in rates at which different components of a mixture move through a porous medium (stationary phase) under the influence of a solvent or gas (mobile or moving phase).
- The technique separates a mixture by distributing its components between one stationary phase and one mobile phase.
- Mobile phase determines the Rᶠ or R factor — dependent on the solubility of components in the given eluent.
| Element | Method / Test | Principle | Observation | Inference |
|---|---|---|---|---|
| Carbon (C) | Copper oxide test | C oxidised to CO₂ by CuO | CO₂ turns lime water milky | Carbon present |
| Hydrogen (H) | Copper oxide test | H oxidised to H₂O by CuO | Anhydrous CuSO₄ turns blue | Hydrogen present |
| Nitrogen (N) | Lassaigne's test | Na + C + N → NaCN → Prussian blue with FeSO₄ + FeCl₃ | Prussian blue colour | Nitrogen present |
| Nitrogen + Sulphur (N+S) | Lassaigne's test | SCN⁻ formed instead of CN⁻ and S²⁻ separately | Blood red colour with FeCl₃ (Fe(SCN)²⁺) | Both N and S present |
| Sulphur (S) only | Sodium nitroprusside test | Na₂S + [Fe(CN)₅NO]²⁻ → coloured complex | Violet colour | Sulphur present |
| Chlorine (Cl) | Lassaigne's test + AgNO₃ | NaCl + AgNO₃ → AgCl↓ | White ppt, soluble in NH₄OH | Chlorine present |
| Bromine (Br) | Lassaigne's test + AgNO₃ | NaBr + AgNO₃ → AgBr↓ | Pale yellow ppt, partially soluble in NH₄OH | Bromine present |
| Iodine (I) | Lassaigne's test + AgNO₃ | NaI + AgNO₃ → AgI↓ | Yellow ppt, insoluble in NH₄OH | Iodine present |
| Halogen (Cl/Br) | Beilstein test | Organic compound on Cu wire in Bunsen flame | Green/blue-green flame | Halogen (Cl or Br) present |
| Phosphorus (P) | Ammonium molybdate test | P oxidised to phosphate → ammonium phosphomolybdate | Canary yellow ppt | Phosphorus present |
Concepts [33]
- Tetravalence of Carbon - Shapes of Organic Compounds
- Complete, Condensed and Bond-line Structural Formulas
- Three-dimensional Representation of Organic Molecules
- Classification of Organic Compounds
- Nomenclature
- IUPAC Nomenclature of Alkanes
- Nomenclature of Organic Compounds having Functional Group(s)
- Nomenclature of Substituted Benzene Compounds
- Isomerism
- Fission of a Covalent Bond
- Nucleophiles and Electrophiles
- Electron Movement in Organic Reactions
- Electron Displacement Effects in Covalent Bonds
- Inductive Effect
- Resonance Structure
- Resonance Effect
- Electromeric Effect (E Effect)
- Hyperconjugation
- Types of Organic Reactions and Mechanisms
- Purification of Organic Compounds
- Sublimation
- Crystallisation Method
- Simple Distillation Method
- Solvent Extraction (Using a Separating Funnel Method)
- Chromatography Method
- Qualitative Analysis of Organic Compounds
- Qualitative Analysis of Organic Compounds - Detection of Other Elements
- Quantitative Analysis of Carbon and Hydrogen
- Quantitative Analysis of Nitrogen
- Quantitative Analysis of Halogens
- Quantitative Analysis of Sulphur
- Quantitative Analysis of Phosphorus
- Quantitative Analysis of Oxygen
