Advertisements
Advertisements
प्रश्न
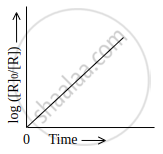
Which of the following graphs is correct for a first order reaction?




Advertisements
उत्तर


Explanation:
The pace of a reaction that is directly proportional to the concentration of the reacting substance is known as a first-order reaction.
A first-order reaction's kinetic equation
k = `2.303/t log [R]^0/([R])`
The Y-axis represents concentration, whereas the X-axis represents time.
`log = [R]^0/([R]) = (kt)/2.303`
`[log = [R]_0/([R])] = [(kt)/2.303](t)`
y = mn
k = `2.303/t log a/(a - x)`
t = `2.303/k log a/(a - x)`
x = `a - a/2`
x = `a/2`
`t_(1/2) = 2.303/k log a/(a - a/2)`
`2.303/k log 2`
`t_(1/2) = 0.693/k`
APPEARS IN
संबंधित प्रश्न
Sucrose decomposes in acid solution into glucose and fructose according to the first order rate law with t1/2 = 3 hours. What fraction of the sample of sucrose remains after 8 hours?
Time required to decompose SO2Cl2 to half of its initial amount is 60 minutes. If the decomposition is a first order reaction, calculate the rate constant of the reaction.
In a pseudo first order hydrolysis of ester in water, the following results were obtained:
| t/s | 0 | 30 | 60 | 90 |
| [A]/mol L−1 | 0.55 | 0.31 | 0.17 | 0.085 |
Calculate the average rate of reaction between the time interval 30 to 60 seconds.
The rate constant for a first order reaction is 60 s−1. How much time will it take to reduce the initial concentration of the reactant to its `1/16`th value?
For the decomposition of azoisopropane to hexane and nitrogen at 543 K, the following data are obtained.
| t (sec) | P(mm of Hg) |
| 0 | 35.0 |
| 360 | 54.0 |
| 720 | 63.0 |
Calculate the rate constant.
A first order reaction is 50% complete in 25 minutes. Calculate the time for 80% completion of the reaction.
Show that the time required for 99.9% completion of a first-order reaction is three times the time required for 90% completion.
The rate constant of a first order reaction is 6.9 × 10–3s–1. How much time will it take to reduce the initial concentration to its 1/8th value?
In a first order reaction the concentration of reactants decreases from 400mol L-1 to 25 mol L-1 in 200 seconds. The rate constant for the reaction is ______.
Observe the graph shown in figure and answer the following questions:
- What is the order of the reaction?
- What is the slope of the curve?
- Write the relationship between k and t1/2 (half life period).
A definite volume of H2O2 undergoing spontaneous decomposition required 22.8 c.c. of standard permanganate solution for titration. After 10 and 20 minutes respectively the volumes of permanganate required were 13.8 and 8.25 c.c. The time required for the decomposition to be half completed is ______ min.
Radioactive decay follows first-order kinetics. The initial amount of two radioactive elements X and Y is 1 gm each. What will be the ratio of X and Y after two days if their half-lives are 12 hours and 16 hours respectively?
The following data were obtained during the decomposition of SO2Cl2 at the constant volume. SO2Cl2 →SO2(g) + Cl2(g)
| Time (s) | Total Pressure (bar) |
| 0 | 0.5 |
| 100 | 0.6 |
Calculate the rate constant of the reaction.
The rate constant for the reaction:
\[\ce{2N2O_{(s)} ->2N2O4_{(g)}}\] is 4.98 × 10-4 s-1.
The order of the reaction is ______.
Write the equation for integrated rate law for a first order reaction.
Show that `t_(1/2)= 0.693/k` for first reaction.
Write the unit of rate constant [k] for the first order reaction.
If the half-life (t1/2) for a first order reaction is 1 minute, then the time required for 99.9% completion of the reaction is closest to ______.
