Advertisements
Advertisements
प्रश्न
Observe the graph shown in figure and answer the following questions:
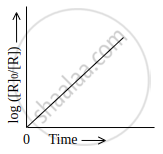
- What is the order of the reaction?
- What is the slope of the curve?
- Write the relationship between k and t1/2 (half life period).
Advertisements
उत्तर
- The order of the reaction is first-order reaction.
- Slope of the curve = `k/2.303`
- The half-life for the first-order reaction is given by t1/2 = `0.693/k`
APPEARS IN
संबंधित प्रश्न
The following data were obtained during the first order thermal decomposition of SO2Cl2 at a constant volume.
\[\ce{SO2Cl2_{(g)} -> SO2_{(g)} + Cl2_{(g)}}\]
| Experiment | Time/s–1 | Total pressure/atm |
| 1 | 0 | 0.5 |
| 2 | 100 | 0.6 |
Calculate the rate of the reaction when total pressure is 0.65 atm.
In a pseudo first order hydrolysis of ester in water, the following results were obtained:
| t/s | 0 | 30 | 60 | 90 |
| [A]/mol L−1 | 0.55 | 0.31 | 0.17 | 0.085 |
Calculate the average rate of reaction between the time interval 30 to 60 seconds.
The rate constant for a first order reaction is 60 s−1. How much time will it take to reduce the initial concentration of the reactant to its `1/16`th value?
A first order reaction takes 20 minutes for 25% decomposition. Calculate the time when 75% of the reaction will be completed.
(Given : log = 2 = 0·3010, log 3 = 0·4771, log 4 = 0·6021)
Which of the following graphs is correct for a first order reaction?




With the help of an example explain what is meant by pseudo first order reaction.
A definite volume of H2O2 undergoing spontaneous decomposition required 22.8 c.c. of standard permanganate solution for titration. After 10 and 20 minutes respectively the volumes of permanganate required were 13.8 and 8.25 c.c. The time required for the decomposition to be half completed is ______ min.
The reaction \[\ce{SO2Cl2(g) -> SO2(g) + Cl2(g)}\] is a first-order gas reaction with k = 2.2 × 10−5 sec−1 at 320°C. The percentage of SO2Cl2 is decomposed on heating this gas for 90 min, is ______%.
The slope in the plot of `log ["R"]_0/(["R"])` Vs. time for a first-order reaction is ______.
Write the equation for integrated rate law for a first order reaction.
