Advertisements
Advertisements
प्रश्न
Which of the following compounds will react with sodium hydroxide solution in water?
विकल्प
\[\ce{C6H5OH}\]
\[\ce{C6H5CH2OH}\]
\[\ce{(CH3)3COH}\]
\[\ce{C2H5OH}\]
Advertisements
उत्तर
\[\ce{C6H5OH}\]
Explanation:
Phenol being more acidic reacts with sodium hydroxide solution in water to give sodium phenoxide which is resonance stabilized.
Alcohols are very weak acids.
\[\ce{C6H5OH + NaOH -> C6H5ONa + H2O}\]
APPEARS IN
संबंधित प्रश्न
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
|\phantom{....................}|\phantom{.........}|\phantom{.............}\\
\ce{OH}\phantom{................}\ce{OH}\phantom{...}\ce{C{_2}H5}\phantom{.........}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH3}\\
|\phantom{.........}|\phantom{...}\\
\ce{OH}\phantom{....}\ce{OH}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{...}\\
\ce{OH}\phantom{.}\\
\end{array}\]
Write the IUPAC name of the following compound:
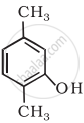
Write the IUPAC name of the following compound:
C6H5 – O – C2H5
How is phenol converted into the following?
Benzene
How is phenol converted into the following?
picric acid
Give reasons Fluoride ion has higher hydration enthalpy than chloride ion.
Give IUPAC names of the following compound:
\[\begin{array}{cc}
\phantom{..}\ce{H}\phantom{...}\ce{CH3}\phantom{.}\ce{H}\phantom{..}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{.}\ce{H}\phantom{...}\ce{OH}\phantom{.}\ce{H}\phantom{.}\\
\end{array}\]
The compound HOCH2 – CH2OH is __________.
The product of acid catalysed hydration of 2-phenylpropene is:
Which of the following gives a positive iodoform test?
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
The correct acidic strength order of the following is:

(I)
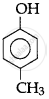
(II)

(III)
Assertion: Addition reaction of water to but-1-ene in acidic medium yields butan-1-ol.
Reason: Addition of water in acidic medium proceeds through the formation of primary carbocation.
Assertion: IUPAC name of the compound
\[\begin{array}{cc}
\ce{CH3 - CH - O - CH2 - CH2 - CH3}\\
|\phantom{....................}\\
\ce{CH3}\phantom{.................}
\end{array}\] is 2-Ethoxy-2-methylethane.
Reason: In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by —OR or —OAr group [where R = alkyl group and Ar = aryl group]
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
How can phenol be converted to aspirin?
The IUPAC name of 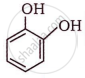 is ______.
is ______.
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
