Advertisements
Advertisements
प्रश्न
What is "rusting"? Describe with a labelled diagram an activity to investigate the conditions under which iron rusts.
Advertisements
उत्तर
When iron is exposed to moist air for a long time, it acquires a coating of a brown flaky substance called rust. This process is known as rusting.
To investigate the conditions under which iron rust, the following activity can be performed:
- Take three test tubes and place clean iron in each of them.
- Label these test tubes A, B and C. Pour some water in test tube A and cork it.
- Pour boiled distilled water in test tube B, add about 1 ml of oil and cork it.
- Put some anhydrous calcium chloride in test tube C and cork it.
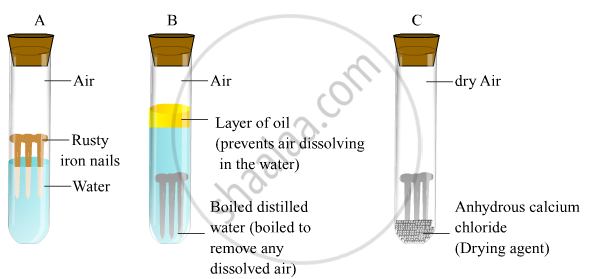
On leaving these test tubes for a few days, it will be observed that iron nails rust in test tube A but they do not rust in test tubes B and C.
In the test tube A, the nails are exposed to both air and water.
In the test tube B, the nails are exposed to only water because the oil will float on water and prevent the air from dissolving in the water.
In the test tube C, the nails are exposed to dry air because anhydrous calcium chloride will absorb the moisture from the air.
Hence, air and water both are essential conditions for rusting.
APPEARS IN
संबंधित प्रश्न
Explain why rusting of iron objects is faster in coastal areas than in deserts.
What is anodising? Give its applications.
What is meant by 'rusting of iron'? With the help of labelled diagrams, describe an activity to find out the conditions under which iron rusts.
Explain why, when a copper object remains in damp air for a considerable time, a green coating is formed on its surface. What is this process known as?
In stainless steel alloy, iron metal is mixed with:
(a) Cu and Cr
(b) Cr and Ni
(c) Cr and Sn
(d) Cu and Ni
Rusting of iron : Fe2O3 : : corrosion of copper : ______
Find the odd one out and give its explanation.
Explain concept with example/explain with the help of a balanced equation.
Corrosion
Give preventive methods by giving examples of corrosion?
Explain the following:
Bubbles are produced when acetic acid is added to a solution of sodium hydrogencarbonate.
