Advertisements
Advertisements
प्रश्न
What is "rusting"? Describe with a labelled diagram an activity to investigate the conditions under which iron rusts.
Advertisements
उत्तर
When iron is exposed to moist air for a long time, it acquires a coating of a brown flaky substance called rust. This process is known as rusting.
To investigate the conditions under which iron rust, the following activity can be performed:
- Take three test tubes and place clean iron in each of them.
- Label these test tubes A, B and C. Pour some water in test tube A and cork it.
- Pour boiled distilled water in test tube B, add about 1 ml of oil and cork it.
- Put some anhydrous calcium chloride in test tube C and cork it.
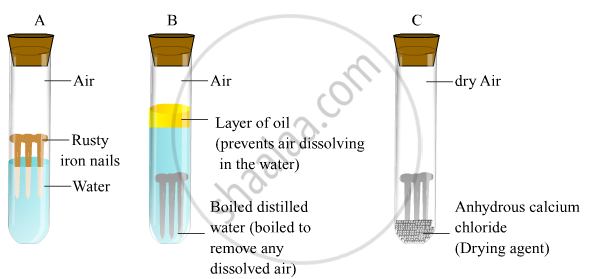
On leaving these test tubes for a few days, it will be observed that iron nails rust in test tube A but they do not rust in test tubes B and C.
In the test tube A, the nails are exposed to both air and water.
In the test tube B, the nails are exposed to only water because the oil will float on water and prevent the air from dissolving in the water.
In the test tube C, the nails are exposed to dry air because anhydrous calcium chloride will absorb the moisture from the air.
Hence, air and water both are essential conditions for rusting.
APPEARS IN
संबंधित प्रश्न
Give reasons:
Platinum, gold and silver are used to make jewellery.
Choose the correct answer from the options given below:
Heating an ore in a limited supply of air or in the absence of air at a temperature just below its melting point is known as
A. Smelting
B. Ore-dressing
C. Calcination
D. Bessemerisation
What is anodising? Give its applications.
Fill in the following blanks with suitable words:
Tiffin boxes are electroplated with .............. but car bumpers are electroplated with ............... to protect them from rusting.
Fill in the following blank with suitable word:
The corrosion of copper produces a .............. coating of basic copper carbonate on its surface
Write the name.
An alloy of copper and tin-
Write scientific reason.
Anodization method is useful for prevention of the corrosion of the aluminium.
State two conditions necessary for rusting of iron.
Marble’s popularity began in ancient Rome and Greece, where white and off-white marble were used to construct a variety of structures, from hand-held sculptures to massive pillars and buildings.

The substance not likely to contain CaCO3 is:
Iron pillar near the Qutub Minar in Delhi is famous for the following facts. Which of these facts is responsible for its long stability?
