Advertisements
Advertisements
प्रश्न
Taking the volume of hydrogen as calculated in Q.19, what change must be made in Kelvin (absolute) temperature to return the volume to 2500 cm3 (pressure remaining constant)?
Advertisements
उत्तर
V1 = 714.29 cm3
P1 = P2 = P
T1 = 273 K
V2 = 2500 cm3
T2= ?
By using Charles's Law,
`("P"_1 "V"_1)/"T"_1 = ("PV"_2)/"T"_2`
Since P is constant, it cancels out.
`"V"_1/"T"_1 = "V"_2/"T"_2`
T2 = `("V"_2 xx "T"_1)/"V"_1`
T2 = `(2500 xx 273)/714.29`
T2 = 3.5 × 273
T2 = 955.5 K
∴ T2 = 3.5 times
∴ The temperature must be increased 3.5 times.
APPEARS IN
संबंधित प्रश्न
Give the assumptions of the kinetic molecular theory.
State the law which is represented by the following graph:
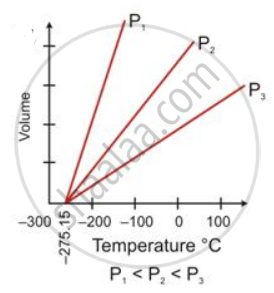
State the following:
Ice point in absolute temperature
Choose the correct answer:
The volume-temperature relationship is given by
What will be the minimum pressure required to compress 500 dm3 of air at 1 bar to 200 dm3 temperature remaining constant?
2 liters of gas is enclosed in a vessel at a pressure of 760 mmHg. If the temperature remains constant, calculate pressure when volume changes to 4 dm3.
It is found that on heating a gas its volume increases by 50% and its pressure decreases to 60% of its original value. If the original temperature was −15°C, find the temperature to which it was heated.
A certain mass of gas occupied 850 ml at a pressure of 760 mm of Hg. On increasing the pressure it was found that the volume of the gas was 75% of its initial value. Assuming constant temperature, find the final pressure of the gas?
The pressure on one mole of gas at s.t.p. is doubled and the temperature is raised to 546 K. What is the final volume of the gas ? [one mole of a gas occupies a volume of 22.4 litres at stp.]
The following question refers to one mole of chlorine gas.
What volume will it occupy at 273°C?
