Advertisements
Advertisements
प्रश्न
Rate law for the reaction \[\ce{A + 2B -> C}\] is found to be Rate = k [A][B]. Concentration of reactant ‘B’ is doubled, keeping the concentration of ‘A’ constant, the value of rate constant will be ______.
विकल्प
the same
doubled
quadrupled
halved
Advertisements
उत्तर
Rate law for the reaction \[\ce{A + 2B -> C}\] is found to be Rate = k [A][B]. Concentration of reactant ‘B’ is doubled, keeping the concentration of ‘A’ constant, the value of rate constant will be the same.
Explanation:
The rate of concentration of a reaction does not depend upon the concentrations of the reactants. Hence, it will remain the same. Even if the equation shows the double concentration level, even the rate concentration doubles so it's the same throughout.
Following with the equation \[\ce{A + 2B -> C}\]
If rate considered as (1) Rate1 = k[A][B]
If rate considered as 2
Then, Rate1 = k[A][2B]
Rate2 = 2Rate1
APPEARS IN
संबंधित प्रश्न
For the first order thermal decomposition reaction, the following data were obtained:

Time / sec Totalpressure / atm
0 0.30
300 0.50
Calculate the rate constant
(Given: log 2 = 0.301, log3 = 0.4771, log 4 = 0.6021)
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{CH3CHO_{(g)} -> CH4_{(g)} + CO_{(g)}}\] Rate = k [CH3CHO]3/2
A reaction is first order in A and second order in B. How is the rate affected when the concentrations of both A and B are doubled?
Write the principle behind the following methods of refining:
Hydraulic washing
Which of the following statement is true for order of a reaction?
Consider a first order gas phase decomposition reaction given below :
\[\ce{A(g) -> B(g) + C(g)}\]
The initial pressure of the system before decomposition of A was pi. After lapse of time ‘t’, total pressure of the system increased by x units and became ‘pt’ The rate constant k for the reaction is given as ______.
The value of rate constant of a pseudo first order reaction ______.
For a complex reaction:
(i) order of overall reaction is same as molecularity of the slowest step.
(ii) order of overall reaction is less than the molecularity of the slowest step.
(iii) order of overall reaction is greater than molecularity of the slowest step.
(iv) molecularity of the slowest step is never zero or non interger.
For which type of reactions, order and molecularity have the same value?
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?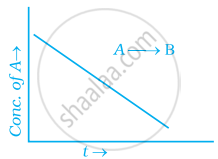
Match the graph given in Column I with the order of reaction given in Column II. More than one item in Column I may link to the same item of Column II.
| Column I | Column II | |
| (i) |  |
|
| (ii) |  |
(a) 1st order |
| (iii) |  |
(b) Zero-order |
| (iv) |  |
Assertion: Order of the reaction can be zero or fractional.
Reason: We cannot determine order from balanced chemical equation.
If the 0.05 molar solution of m+ is replaced by a 0.0025 molar m+ solution, then the magnitude of the cell potential would be
The half-life period of a. substance in a certain enzyme catalysed reaction is 138 s. The time required for the concentration of the substance to fall from 1.28 mol–1 to 0.04 mg L–1 is
The number of molecules of the reactants taking part in a single step of the reaction is indicative of ______.
The following data was obtained for chemical reaction given below at 975 K.
\[\ce{2NO(g) + 2H2(g) -> N2(g) + 2H2O(g)}\]
| [NO] | [H2] | Rate | |
| Mol L-1 | Mol L-1 | Mol L-1 s-1 | |
| (1) | 8 × 10-5 | 8 × 10-5 | 7 × 10-9 |
| (2) | 24 × 10-5 | 8 × 10-5 | 2.1 × 10-8 |
| (3) | 24 × 10-5 | 32 × 10-5 | 8.4 × 10-8 |
The order of the reaction with respect to NO is ______. (Integer answer)
For a chemical reaction starting with some initial concentration of reactant At as a function of time (t) is given by the equation,
`1/("A"_"t"^4) = 2 + 1.5 xx 10^-3` t
The rate of disappearance of [A] is ____ × 10-2 M/sec when [A] = 2 M.
[Given: [At] in M and t in sec.]
[Express your answer in terms of 10-2 M /s]
[Round off your answer if required]
A reaction is second order with respect to a reactant. How is the rate of reaction affected if the concentration of the reactant is reduced to half?
